Sourcing Disclosure: Licensed Bangladesh Global Sourcing Specialist facilitating global access under Named Patient Regulations.
Expert-reviewed clinical data for prescription-only medicine.
Evolving Production Standards and Accessibility in Generic Oncology Therapeutics
We have moved beyond the era where cancer care was defined strictly by high-cost, exclusive branded drugs accessible only to a small percentage of the global population. Today, the focus has shifted toward the democratization of treatment through scientific advancement and manufacturing efficiency. This shift is led by key production trends in the generic pharmaceutical sector that are making the most advanced treatments safer, highly portable, and significantly more affordable. For patients and caregivers, understanding these trends is not just about understanding the market; it is about recognizing the new clinical tools available for survival. This guide provides a technical analysis of the engineering and regulatory advancements defining modern generic oncology therapeutics.
1. The Proliferation of Oral Targeted Therapies
One of the most visible changes in patient care is the transition from hospital-based intravenous (IV) infusions to home-based oral medications. Historically, chemotherapy required patients to spend hours in a clinical setting, adding significantly to the cost and psychological burden of care. Today, the rise of oral generic oncology therapeutics—specifically Tyrosine Kinase Inhibitors (TKIs)—has changed the standard protocol for many solid tumors.
The production of these oral generics relies on advanced pharmaceutical engineering. Manufacturers optimize the crystallization processes of these molecules to ensure they are completely stable in pill form. Generic TKIs for lung, breast, and blood cancers are now produced with such high precision that they achieve the exact same dissolution kinetics as their branded predecessors. For the patient, this means the ability to manage a serious illness at home, reducing hospital-related costs and allowing for a return to a more normal daily routine.
2. Biosimilars and Advanced Immunotherapy
While traditional generic drugs are chemical copies of simple molecules, the most advanced cancer drugs today are “biologics”—complex medicines synthesized from living cells. The non-innovator versions of these therapies are classified as biosimilars.
Producing a biosimilar is significantly more complex than producing a standard generic. It requires advanced bioreactors and precise control over the cellular environment, as even a microscopic change in the growth medium can alter the final protein structure. Consequently, biosimilars are not considered exact “generics,” but rather highly similar therapeutics with no clinically meaningful differences. By using analytical tools like high-resolution mass spectrometry, manufacturers can prove functional parity with the original biologic. This is particularly important for treatments like monoclonal antibodies (mAbs), where the availability of biosimilars has reduced prices by up to 50%, making immunotherapy reachable for millions of previously excluded patients.
3. From Batch to Continuous Manufacturing
A major technical trend driving quality and lowering costs is the shift from “Batch Manufacturing” to “Continuous Manufacturing.” Traditionally, medicines were made in separate steps, which left room for variation between runs. Leading oncology labs have now moved to continuous production lines where raw materials are fed in at one end and the final tablets come out the other in a single, uninterrupted flow.
As an engineering concept, continuous manufacturing offers several advantages for generic oncology therapeutics:
- Real-Time Quality Control: Sensors monitor the chemical composition continuously. If the purity drops even by a fraction of a percent, the system automatically corrects it or diverts the sub-standard material.
- Efficiency: It reduces chemical waste and energy consumption, which directly lowers the production cost.
- Scalability: Manufacturers can respond faster to global drug shortages, ensuring that cancer patients never have to miss a dose due to supply chain failures.
4. Artificial Intelligence in Bioequivalence and Drug Design
Artificial Intelligence (AI) is now a fundamental tool in the generic pharmaceutical industry, actively used to speed up the development of bioequivalent medicines. Instead of relying solely on extended laboratory trials, researchers use in-silico modeling—computer simulations that predict how a generic molecule will dissolve and absorb in the human body.
Algorithms analyze extensive pharmacokinetic data to optimize the formulation of a generic pill. This helps manufacturers choose the perfect combination of inactive ingredients (excipients) to ensure the generic drug matches the branded version’s performance. This computational efficiency shortens the development time, allowing affordable versions of high-tech medicines to reach the market much faster after a patent expires.
5. Serialization and Supply Chain Security
As the generic oncology market has grown, the industry has implemented advanced serialization and blockchain technology to secure the supply chain. Every box of high-quality generic oncology medicine now follows global GS1 serialization standards.
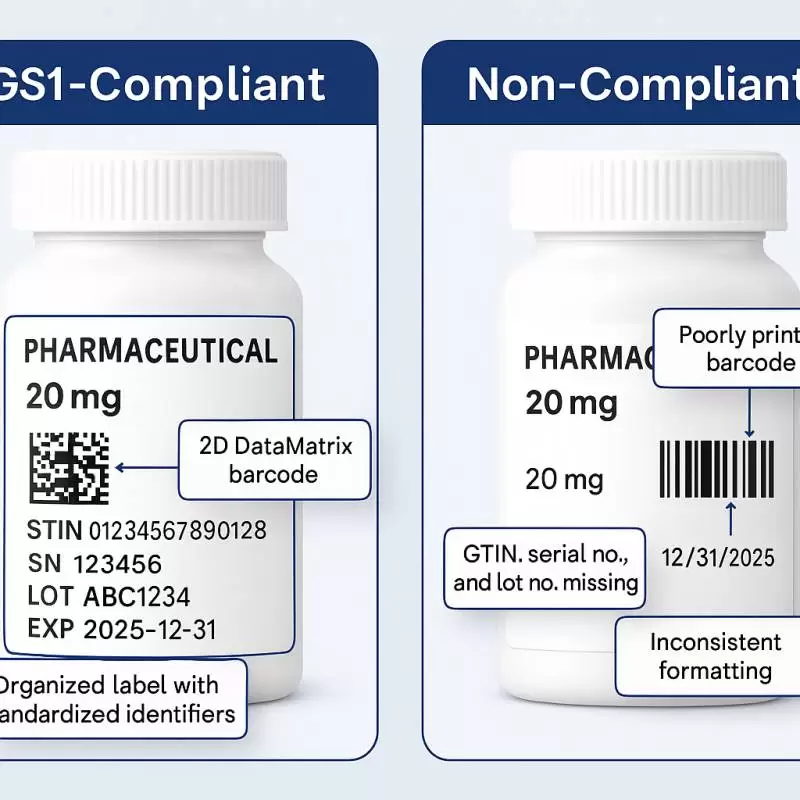
Each package is assigned a unique, encrypted 2D DataMatrix barcode. When a pharmacy receives the medicine, or when a patient scans the box with a smartphone, they can trace the drug’s journey from the exact manufacturing plant to the shelf. While counterfeiting remains a global challenge, this “track and trace” infrastructure provides critical, verifiable safeguards against sub-standard therapeutics, offering patients digital proof of their medication’s authenticity.

6. Pharmacogenomics and Personalized Generic Selection
Clinical oncology has moved away from a “one-size-fits-all” approach. The science of pharmacogenomics—studying how your genes affect your response to drugs—is now directly applied to the selection of generic oncology therapeutics. Simple genomic assays can predict if a patient possesses the specific liver enzymes required to effectively metabolize a targeted therapy, or if they are at high risk for severe side effects.
This personalized approach allows oncologists to select the right generic brand or specific release profile for each individual. It increases the success rate of generic treatments, reduces the physical toll of “trial and error” prescribing, and ensures the patient gets the maximum benefit from their affordable medication right from the start.
7. Sustainable Green Chemistry in Manufacturing
Environmental sustainability is increasingly recognized as a core metric of pharmaceutical quality. The production of complex oncology drugs traditionally involved harsh solvents and high energy usage. The current industry standard is shifting rapidly toward “Green Chemistry,” where manufacturers use biological catalysts and water-based solvents to synthesize drug molecules.
Green chemistry often results in a purer final product with fewer toxic byproducts. For the generic industry, it also reduces the cost of hazardous waste management and raw materials, further driving down the price of the medicine. Patients are increasingly prioritizing therapeutics produced with a commitment to both human health and environmental safety.
8. Direct-to-Patient (DTP) Distribution and Transparency
Historically, a long chain of distributors, wholesalers, and retail pharmacies added multiple layers of markups to the price of cancer care. The emergence of the Direct-to-Patient (DTP) distribution model is dismantling these barriers.
Specialized digital platforms now allow patients to source their generic oncology medications directly from authorized primary distributors. This model strips away intermediary markups, substantially lowering the financial burden on the patient. Furthermore, authoritative platforms provide unprecedented transparency, offering direct access to clinical trial results, GMP certifications, and expert medical reviews. This democratization of data empowers the patient to make highly informed treatment decisions.
9. Global Quality Harmonization
In the past, the quality of a generic drug could vary depending on its country of origin. Today, the pharmaceutical industry operates under a framework of “Global Quality Harmonization.” Driven by the rigorous standards of the International Council for Harmonisation (ICH), the regulatory requirements for generic oncology therapeutics are highly uniform across major global markets.
A high-quality manufacturing facility in South Asia now follows the exact same Current Good Manufacturing Practices (cGMP) as a lab in Europe or North America. This harmonization has built a global level of trust, allowing patients to access medications from international manufacturing hubs with the confidence that the chemical purity and clinical efficacy are identical to what they would receive in any major Western cancer center.
10. Conclusion: Precision and Accessibility
The convergence of oral targeted therapies, biosimilars, continuous manufacturing, and digital supply chain security has created a reality where high-quality cancer care is no longer a luxury. Generic oncology is the driving force of this transformation. By stripping away unnecessary costs and embracing high-tech engineering efficiencies, the generic pharmaceutical sector ensures that the most effective molecules reach the patients who need them most.
As a patient or caregiver, understanding these manufacturing and regulatory standards allows you to navigate your treatment with authority, choosing your medicine based on verifiable scientific fact.
Medical Disclaimer: The information provided in this article is for educational and awareness purposes only and is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Every patient’s medical situation is unique. Decisions regarding the use of new trends, technologies, or generic oncology therapeutics must be made in consultation with a qualified oncologist or a registered medical professional. The clinical application of pharmacogenomics or DTP distribution should be handled under the guidance of your treating physician and a licensed pharmacist. Medically Reviewed by Dr.Salma Elreedy