Sourcing Disclosure: Licensed Bangladesh Global Sourcing Specialist facilitating global access under Named Patient Regulations.
Expert-reviewed clinical data for prescription-only medicine.
The Clinical Integration of Generic Oncology Medicines
Article Overview
The news of a cancer diagnosis is a life-changing event that brings immediate emotional and practical challenges. Beyond the medical complexities of the disease, the cost of treatment often becomes a primary concern for patients and their families. In the field of oncology, the price of medication can be so high that it creates a barrier to receiving consistent care. This is why understanding generic oncology medicines is essential. These medications offer a way to receive high-quality, life-saving treatment at a price that is manageable. This guide explains the science, safety, and economics of generic cancer drugs in 2026 to help you make informed decisions about your health.
1. The Core Definition of a Generic Medicine
A generic medicine is a pharmaceutical product that is designed to be the same as an already marketed brand-name drug. In 2026, the standards for these medications are stricter than ever. A generic drug must match the original branded version in several key areas: its active ingredient, its dosage form, its strength, and its route of administration (how it is taken).
When we talk about oncology, we are dealing with high-potency drugs that target specific cellular functions. A generic oncology drug is created once the patent of the original “innovator” drug expires. At this point, other manufacturers are legally allowed to produce the same chemical molecule. This molecule is known as the Active Pharmaceutical Ingredient (API). The API is the part of the medicine that actually performs the work of attacking cancer cells or stopping their growth.
2. The Science of Molecular Identity and Bioequivalence
In 2026, the primary standard for a generic oncology drug is Bioequivalence. This is a scientific term that means the generic drug works in the human body in the same way and at the same rate as the brand-name drug.
The Chemical Blueprint
At the molecular level, there is no difference between the API of a high-quality generic and a branded drug. If the brand-name drug uses a specific molecule like Imatinib or Erlotinib, the generic version must use the exact same molecular structure. For someone with a background in science, this is easy to understand: a molecule is defined by its chemical bonds and atoms, and these do not change regardless of which company manufactures them.
Testing for Efficacy
To prove bioequivalence, generic manufacturers must perform clinical tests. They measure how quickly the drug enters the bloodstream and how long it stays there. They focus on three main metrics:
- Cmax: The peak concentration of the drug in the blood.
- Tmax: The time it takes to reach that peak.
- AUC: The total exposure of the drug to the body over time.
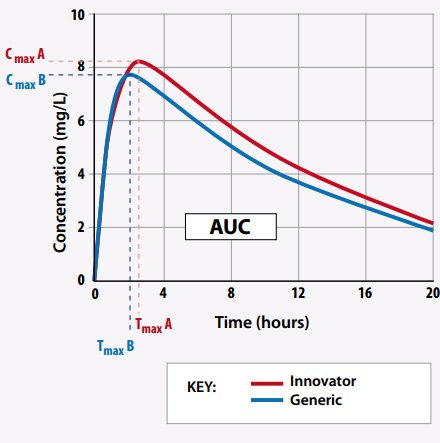
For a drug to be approved in 2026, these numbers must be virtually identical to the branded version. This ensures that the patient receives the same clinical benefit, whether they choose the expensive brand or the affordable generic.
3. Why Generic Oncology Medicines are More Affordable
A common misunderstanding is that generic drugs are cheaper because they use “cheaper” or “lower quality” ingredients. This is factually incorrect. The lower price of a generic drug is a result of a different business model, not a lower standard of science.
No Research & Development (R&D) Costs
The original manufacturer of a drug spends years and billions of dollars on the initial discovery of the molecule. They have to pay for the failures of thousands of other molecules that didn’t work. The high price of the branded drug is designed to recover these initial costs. Generic manufacturers do not have to “re-invent the wheel.” They start with a molecule that has already been proven to work.
Abbreviated Clinical Trials
Generic companies do not need to repeat the massive, multi-year Phase III clinical trials that the original company performed to prove the drug’s safety for the first time. Since the original company has already established that the molecule is safe and effective, the generic company only needs to prove that their version is bioequivalent. This saves hundreds of millions of dollars in development costs.

Market Competition
In 2026, the pharmaceutical market operates on the principle of competition. When a patent expires and five different companies start making the same drug, they must compete on price to attract hospitals and patients. This competition naturally drives the cost down. It is a win for the patient, as the cost of treatment can drop by 60% to 90% without any reduction in the quality of the drug.
4. Understanding Inactive Ingredients (Excipients)
While the Active Pharmaceutical Ingredient (API) in a generic drug must be identical to the brand, the Inactive Ingredients (called excipients) can be different. Excipients are the binders, fillers, and coatings that hold the pill together or help it dissolve.
In 2026, all excipients used in oncology must be proven safe. Some patients might be sensitive to a specific dye or filler used in a generic, just as they might be sensitive to one used in a branded drug. However, these inactive ingredients do not change how the medicine fights the cancer. If a doctor prescribes a generic, they have verified that the inactive ingredients are safe for human consumption and meet international pharmaceutical standards.
5. Quality Control: The GMP Standard in 2026
Quality is the most important factor in oncology. Because cancer drugs are highly potent, even a small error in manufacturing can be dangerous. In 2026, high-quality generic oncology drugs are produced in facilities that follow Good Manufacturing Practices (GMP).
GMP is a system of rules that ensures products are consistently produced and controlled according to quality standards. It covers everything from the purity of the air in the factory to the cleanliness of the machines and the training of the staff. When a generic drug is manufactured in a GMP-certified facility, it means every batch is tested for:
- Purity: To ensure there are no contaminants.
- Strength: To ensure every pill has the exact dose written on the box.
- Stability: To ensure the drug remains effective until its expiration date.
For a website like genericoncology.com, the focus is always on these standards. We rely on data from these high-tech labs to ensure that the medicines discussed are produced at the highest international level.
6. Addressing Common Myths about Generic Cancer Drugs
Even in 2026, several myths about generics continue to circulate. It is important to look at the facts.
Myth: Branded drugs work faster than generics.
Fact: Because they are bioequivalent, they dissolve and enter the bloodstream at the same rate. There is no scientific reason for a brand-name drug to work faster than its generic equivalent.
Myth: Generics are made in “dirty” factories.
Fact: Many branded companies actually use the same factories to produce their drugs as generic companies. Every factory, regardless of who owns it, must pass the same rigorous safety inspections by health authorities.
Myth: Generics cause more side effects.
Fact: Side effects in oncology are usually caused by the Active Pharmaceutical Ingredient (API). Since the API is the same in both, the side effect profile is generally the same. On very rare occasions, a patient might have a mild reaction to an inactive filler, but this is not a common occurrence.
7. The Importance of Medical Review
In the field of oncology, information is a form of care. However, not all information on the internet is reliable. In 2026, the rise of “digital health” means that many websites provide medical information without expert verification.
At genericoncology.com, we emphasize the role of the Medical Reviewer. A medical reviewer is a qualified specialist—often an oncologist or a specialized pharmacist—who checks the accuracy of every clinical statement. This ensures that when you read about a generic drug, you are looking at data that is scientifically sound. This expert oversight is a core part of building trust and ensuring patient safety.
8. Managing Your Treatment Plan with Generics
If you are considering switching to a generic oncology medication, the most important step is to communicate with your medical team.
- Ask your oncologist: “Is there a reputable generic version of my prescribed medication?”
- Check the source: Ensure the generic is coming from a well-known manufacturer with a track record of quality.
- Monitor your progress: As with any medication, keep a diary of how you feel. Your doctor will monitor your blood work and tumor markers to ensure the treatment is working exactly as intended.
In 2026, most oncologists are very supportive of high-quality generics because they know that affordable medicine leads to better treatment adherence. A patient who can afford their medicine is much more likely to complete their full course of treatment, leading to a better clinical outcome.
9. The Global Impact of Generic Oncology
Generic drugs are not just about saving money for an individual; they are about the sustainability of healthcare systems worldwide. As the number of cancer cases increases, no country can afford to treat everyone using only branded medications.
Generic oncology medicines democratize healthcare. They ensure that the latest scientific breakthroughs are not just for the wealthy, but are accessible to everyone. In 2026, manufacturers in South Asia and other global hubs are producing world-class generics that are being used in clinics around the world. This is a vital part of the global effort to make cancer care a human right rather than a financial privilege.
10. Conclusion and Forward Thinking
The year 2026 has brought us to a point where the quality of generic oncology is higher than it has ever been. By stripping away the high costs of marketing and initial research, generic drugs focus on what truly matters: the active molecule that fights the disease. Choosing a generic drug is a scientifically sound, responsible, and effective way to manage cancer treatment.
As you manage your health journey, remember that information is your strongest tool. By understanding the science of bioequivalence, the standards of GMP manufacturing, and the economics of the pharmaceutical industry, you can choose your treatment with confidence. The focus should always be on quality, safety, and clinical results. Generic oncology medicines deliver all three, ensuring that you can focus on your recovery without the added burden of unmanageable debt.
Medical Disclaimer: The information provided in this article is for educational and awareness purposes only. It is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Every patient’s medical situation is unique. Decisions regarding cancer treatment, including the choice between branded and generic medications, must always be made in consultation with a qualified oncologist or a registered medical professional. Never disregard professional medical advice or delay seeking it because of something you have read on this website.