Sourcing Disclosure: Licensed Bangladesh Global Sourcing Specialist facilitating global access under Named Patient Regulations.
Expert-reviewed clinical data for prescription-only medicine.
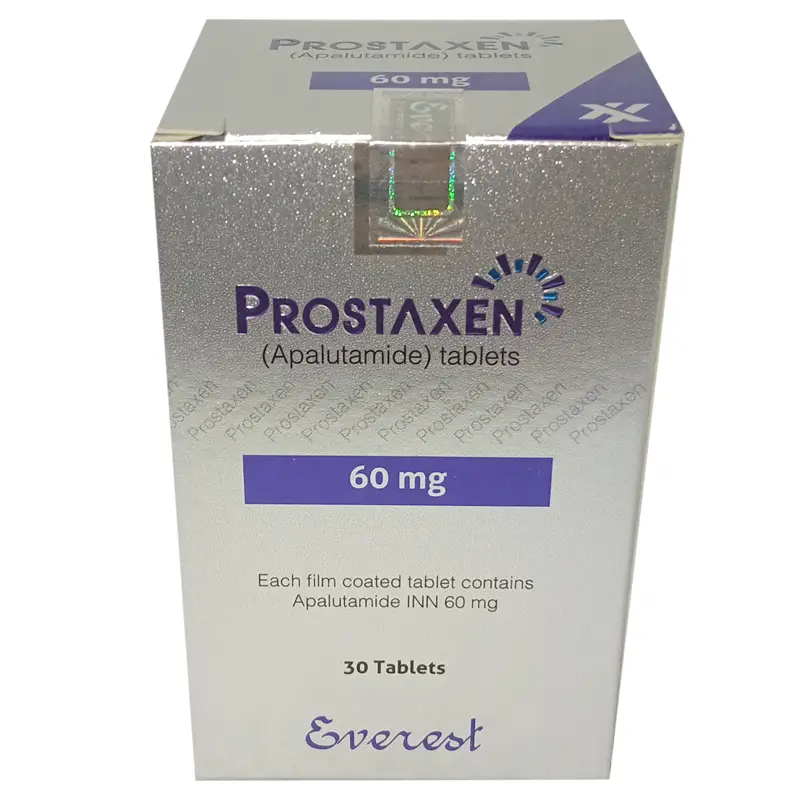
Prostaxen 60 mg Tablet (Apalutamide): Clinical Data, Safety Protocols, & Global Price
- Used for: The treatment of adult men with nmCRPC & mCSPC.
- Availability: In Stock
- Shipping: Express Global Shipping (7-14 days depending on region).
- Requirement: Valid prescription from a licensed healthcare provider required.
✓ WHO GMP Certified
✓ Reviewed By Medical Expert
✓ Batch Examined in Lab
Need Patient Access Support?
Our team provides verified global sourcing assistance to help you navigate international shipping and prescription requirements safely.
Prostaxen 60 mg is a potent, next-generation non-steroidal androgen receptor (AR) inhibitor. It is specifically indicated for the treatment of adult men with Non-Metastatic Castration-Resistant Prostate Cancer (nmCRPC) and Metastatic Castration-Sensitive Prostate Cancer (mCSPC). By selectively binding to the ligand-binding domain of the androgen receptor, Prostaxen inhibits AR nuclear translocation and DNA binding, effectively halting the cellular proliferation signaled by androgens. This targeted mechanism significantly delays the progression to metastatic disease and improves overall survival in advanced prostate cancer populations.
Commentary by Dr. Salma Elreedy, Board-Certified Oncologist
“In the traditional management of prostate cancer, we often faced a ‘therapeutic ceiling’ once a patient developed resistance to primary androgen deprivation therapy (ADT). The transition from nmCRPC to metastatic disease was a period of high anxiety for both clinicians and patients. The advent of second-generation AR inhibitors like Apalutamide represents a paradigm shift. We are no longer just lowering testosterone; we are fundamentally blocking the receptor’s ability to drive malignancy.
In my practice, the clinical utility of Apalutamide lies in its ability to extend Metastasis-Free Survival (MFS) while maintaining a high quality of life. However, managing the unique side-effect profile—specifically the risk of falls and skin rash—requires proactive patient education. The introduction of high-quality generic equivalents like Prostaxen from Everest Pharmaceutical is a vital development. It ensures that this high-efficacy targeted therapy remains accessible to a global patient base, removing the financial toxicity that often accompanies long-term oncological care.“
FDA Approved Indications, Dosage & Adverse Event Management, Clinical Efficacy, and Safety Protocols
Precise Indications & Biomarker Requirements
Treatment with Prostaxen requires a confirmed diagnosis of prostate cancer in specific clinical states:
- nmCRPC: Patients must have non-metastatic disease as confirmed by conventional imaging (CT/MRI/Bone Scan) and a rising Prostate-Specific Antigen (PSA) despite castrate levels of testosterone (< 50 ng/dL).
- mCSPC: Patients with metastatic disease who still respond to testosterone suppression.
- Companion Diagnostic: No specific genetic biomarker (like BRCA) is required, but a documented castrate-resistant status or metastatic sensitivity is mandatory.
Mechanism of Action & Pharmacokinetics (ADME)
Apalutamide works via triple-action AR inhibition: blocking androgen binding, preventing AR nuclear translocation, and inhibiting AR-mediated DNA transcription.
- Absorption: Median Tmax is 2 hours. It can be taken with or without food.
- Distribution: 96% protein bound, primarily to albumin.
- Metabolism: Metabolized primarily by CYP3A4 and CYP2C8 to form N-desmethyl apalutamide (an active metabolite).
- Excretion: 65% eliminated in urine; 24% in feces.
- Half-life: Approximately 3 days at steady state.
Dosage & Adverse Event (AE) Management
The standard dose of Prostaxen is 240 mg (four 60 mg tablets) administered orally once daily. Patients should also receive a gonadotropin-releasing hormone (GnRH) analog concurrently or have had a bilateral orchiectomy. Prostaxen 60 mg tablets should be swallowed whole. Do not crush, chew, or split the tablets.
| Toxicity Type | Severity (Grade) | Clinical Protocol & Dose Modification |
|---|---|---|
| Seizures | Any Grade | Permanently discontinue Prostaxen. Do not re-challenge. |
| Skin Rash | Grade 3 (Severe) | Interrupt treatment. Apply topical steroids or antihistamines. Resume at same or reduced dose (180 mg or 120 mg) when Grade ≤ 1. If the rash returns after dose reduction, the drug should be discontinued. |
| Falls / Fractures | Any | Evaluate for bone-modifying agents (e.g., denosumab). Ensure Vitamin D and Calcium supplementation. |
| Hypothyroidism | Any Grade | Monitor TSH every 4 months. Initiate thyroid replacement therapy as clinically indicated. |
| Other Toxicities | Grade 3 or higher | Interrupt until Grade ≤ 2. Resume at the same dose or reduce by 60 mg if recurrence occurs. |
Clinical Efficacy & Real-World Data
The clinical profile of Apalutamide was solidified in two landmark Phase 3 trials:
- SPARTAN (nmCRPC): Demonstrated a median Metastasis-Free Survival (MFS) of 40.5 months vs. 16.2 months in the placebo group (Hazard Ratio: 0.28).
- TITAN (mCSPC): Showed a 33% reduction in the risk of death (Hazard Ratio: 0.67) and significantly improved Radiographic Progression-Free Survival (rPFS).
Real-World Evidence (RWE): Post-marketing observations confirm that Apalutamide maintains its efficacy in “real-world” elderly populations, though there is a slightly higher incidence of fatigue and falls compared to clinical trial cohorts, emphasizing the need for geriatric assessment during treatment.
Drug-Drug Interaction (DDI) Matrix
Apalutamide is a strong inducer of CYP3A4 and CYP2C19, and a weak inducer of CYP2C9.
| Interaction Category | Agent Examples | Clinical Recommendation |
|---|---|---|
| CYP3A4 Substrates | Midazolam, Fentanyl, Quinidine | Apalutamide induces CYP2C9; expect a decrease in S-warfarin exposure leading to a decreased INR. Perform frequent INR monitoring and adjust Warfarin dosage as required. |
| Anticoagulants | Warfarin | Potential for decreased INR. Monitor Prothrombin Time/INR closely. |
| Strong CYP3A4/2C8 Inhibitors | Itraconazole, Gemfibrozil | No initial dose adjustment, but monitor for increased Apalutamide toxicity. |
| UGT Substrates | Irinotecan | Apalutamide may decrease exposure to UGT substrates. |
Precautions & Special Populations
- Embryo-Fetal Toxicity: Prostaxen is not indicated for use in females. It can cause fetal harm. Males with female partners of reproductive potential must use effective contraception during treatment and for 3 months after the last dose.
- Ischemic Heart Disease: Increased risk observed in trials. Monitor for signs of cardiac ischemia and optimize management of cardiovascular risk factors (BP, lipids).
- Renal/Hepatic Impairment: No dose adjustment is needed for mild-to-moderate impairment. Use with caution in severe renal impairment or severe hepatic impairment (Child-Pugh C).
- Storage: Store at 20°C to 25°C (68°F to 77°F). Protect from light and moisture by keeping tablets in the original bottle.
Manufacturing Quality Assurance, Named Patient Global Access, & Brand Comparison
Everest Pharmaceuticals Manufacturing Insights
Everest Pharmaceutical utilizes advanced synthesis and tableting technologies to produce Prostaxen. The facility is WHO-GMP and ISO 9001:2015 certified, ensuring that every batch undergoes rigorous dissolution testing and bioequivalence studies to match the kinetic profile of the innovator brand. High-Performance Liquid Chromatography (HPLC) is standard for all API verification processes.
Global Access via Named Patient Program (NPP)
Patients can access Prostaxen through the Named Patient Program (NPP), which facilitates the legal importation of medications for personal use:
- Oncology Consultation: Obtain a valid prescription from a registered oncologist.
- Letter of Medical Necessity (LMN): Documentation outlining the clinical need for Apalutamide.
- Permit Acquisition: Apply for a personal import license from the local Drug Regulatory Authority (e.g., DGDA in Bangladesh).
- Logistics: Importation via certified pharmaceutical exporters using temperature-controlled shipping.
Everest (Prostaxen) vs Innovator (Erleada)
| Feature | Innovator (Erleada) | Prostaxen (Everest) |
|---|---|---|
| Active Ingredient | Apalutamide | Apalutamide |
| Dosage Strength | 60 mg | 60 mg |
| API Purity | Reference Standard | Ph. Eur. / USP Grade |
| Indications | nmCRPC, mCSPC | nmCRPC, mCSPC |
| Manufacturing | Janssen-Cilag | Everest Pharma |
People Also Want to Know
What is the price of Prostaxen 60 mg (Apalutamide) and how does it compare to Erleada?
The price of Prostaxen 60 mg typically represents a 60% to 80% reduction in cost compared to the innovator brand, Erleada. In the management of advanced prostate cancer, “financial toxicity” is a documented clinical barrier that leads to treatment non-adherence.
By utilizing Prostaxen, patients can access the identical active pharmaceutical ingredient (Apalutamide) at a sustainable price point. This cost-efficiency is achieved through Everest Pharmaceutical’s streamlined regional manufacturing under WHO-GMP standards, rather than a reduction in chemical purity or clinical efficacy. For specific pricing and international shipping logistics through the Named Patient Program, a valid medical prescription is required to generate a formal quotation.
Can Prostaxen be taken without food?
Yes, Prostaxen can be taken once daily with or without food, but it should be taken at the same time every day to maintain steady plasma levels.
Is Prostaxen 60 mg a type of chemotherapy?
No, it is a targeted hormone therapy (AR inhibitor). It works differently than cytotoxic chemotherapy by blocking the hormonal signals that allow cancer to grow.
What should I do if I miss a dose of Prostaxen?
If a dose is missed, take it as soon as possible on the same day. Resume the regular schedule the next day. Do not take extra tablets to make up for a missed dose.
Why is there a risk of seizures with Apalutamide?
Apalutamide may cross the blood-brain barrier and inhibit GABA receptors at high concentrations. While rare, patients with a history of seizures should be monitored or use alternative therapies.
How long must I use Prostaxen?
Treatment is generally continued until disease progression or the development of unacceptable toxicity, as determined by your oncologist.