Sourcing Disclosure: Licensed Bangladesh Global Sourcing Specialist facilitating global access under Named Patient Regulations.
Expert-reviewed clinical data for prescription-only medicine.
Storing and Handling Oral Oncology Medications at Home
Cancer treatment increasingly relies on oral targeted therapies administered at home rather than intravenous hospital infusions. While this transition offers patients more independence, it transfers the responsibility of medication management directly to the patient and their caregivers.
Unlike standard prescription drugs, oral oncology medications are highly potent, sensitive to environmental changes, and often cytotoxic. Once these medications leave the controlled environment of a pharmacy, improper storage or handling can compromise the treatment’s effectiveness and pose serious health risks to other household members.
1. Chemical Stability: The Critical Role of Temperature Control
Temperature regulation is the most critical factor in maintaining the therapeutic efficacy of oral oncology medications. Advanced targeted therapies have highly specific chemical structures that possess a narrow stability range. Exposure to temperatures outside this designated range can accelerate chemical degradation, effectively rendering the active pharmaceutical ingredients sub-therapeutic or entirely ineffective.
1.1 Defining “Controlled Room Temperature”
The majority of oral oncology tablets are formulated to be stored at “controlled room temperature.” Pharmacological guidelines strictly define this as 20°C to 25°C (68°F to 77°F).
Particularly in warmer, tropical climates where ambient household temperatures can easily exceed 30°C (86°F) during the day, patients cannot rely on general room air to maintain drug stability. It is vital to designate a consistently cool, shaded storage area. Medications must be kept away from direct sunlight, heating vents, and heat-emitting electronic appliances.
1.2 High-Risk Storage Zones: The Kitchen and Bathroom
The two most common household locations for medication storage are strictly contraindicated for oncology drugs:
- The Kitchen: Experiences severe, unpredictable temperature fluctuations and ambient heat from ovens and stovetops.
- The Bathroom: Generates high levels of steam, heat, and humidity from showers.
The combination of thermal stress and environmental moisture in these rooms can prematurely break down enteric coatings and dissolve capsules, even when the medication remains sealed in its original blister packaging.
1.3 Protocols for Refrigerated (Cold-Chain) Medications
Certain liquid oncology formulations, specialized suspensions, and biologic agents require continuous “cold-chain” storage. This typically mandates a strict temperature range between 2°C and 8°C (36°F and 46°F). To manage refrigerated drugs safely at home, adhere to the following protocols:
- Avoid the Refrigerator Door: Never store oncology medications in the door compartments. The temperature here fluctuates violently every time the appliance is opened.
- Optimal Placement: Store the medication in its original carton on a middle shelf, positioned toward the back where the internal temperature remains the most consistent.
- Prevent Freezing: Ensure the medication carton does not directly touch the back wall or cooling element of the refrigerator. Accidental freezing can cause irreversible degradation of the drug’s molecular structure.

2. Environmental Protection: Mitigating Light and Moisture Exposure
Light and moisture are significant factors in the premature degradation of pharmaceutical compounds. Many oral oncology medications, including specialized generic formulations, utilize sophisticated enteric or extended-release coatings. These coatings are engineered to control the precise site and rate of drug absorption within the gastrointestinal tract. Premature environmental exposure can compromise these delivery mechanisms, leading to sub-optimal therapeutic outcomes.
2.1 The Integrity of Original Packaging
Maintaining oncology medications in their original manufacturer packaging is a critical safety and quality requirement. Pharmaceutical packaging is specifically engineered to serve as a high-barrier shield against environmental contaminants.
- Blister Packaging: Many targeted therapies are dispensed in individual foil-sealed blister packs. This design provides an airtight seal for each unit dose, preventing exposure to atmospheric humidity.
- Hygroscopic Properties: Modern small-molecule inhibitors are frequently “hygroscopic,” meaning they actively attract and absorb water molecules from the air. If a hygroscopic tablet is removed from its foil or original bottle and placed in a standard plastic pill organizer, it can begin to degrade within minutes, potentially altering the drug’s chemical stability.

2.2 Managing Photosensitivity
A high percentage of oncology agents are “photosensitive,” meaning they undergo chemical breakdown when exposed to ultraviolet (UV) or ambient light. This degradation can produce inactive or even toxic byproducts.
- Opaque and Amber Containers: Medications that are sensitive to light are typically dispensed in light-resistant amber bottles or opaque secondary packaging. To ensure the drug remains stable, do not transfer these medications into clear containers for the sake of convenience.
- The “Double-Shield” Protocol: Clinicians recommend keeping light-sensitive bottles inside their original cardboard cartons. This provides an additional layer of UV protection. When administering a dose, remove the medication from its container quickly and reseal the bottle or box immediately to minimize cumulative light exposure.
3. Household Biosafety: Establishing a “Chemo-Safe” Environment
Oral oncology medications are clinically classified as hazardous drugs. While these agents are therapeutic for the patient, they are cytotoxic—meaning they are designed to inhibit or kill rapidly dividing cells. In healthy individuals, accidental exposure can interfere with normal cellular replication. This risk is most acute for developing fetuses, nursing infants, and young children, where cellular division is at its peak.
To maintain a safe residential environment, patients and caregivers must implement rigorous biosafety protocols.
3.1 Mitigating Accidental Ingestion
The potency of modern antineoplastic agents means that even a single accidental dose can result in severe toxicity. Standard safety practices must be strictly enforced:
- Secured Storage: Medications must be stored in a dedicated, elevated, and locked cabinet. Do not store oncology drugs in common areas where they can be confused with standard over-the-counter medications.
- Point-of-Use Safety: Avoid leaving medication strips or bottles on bedside tables or kitchen counters. For children, brightly colored capsules can be mistaken for confectionery; therefore, all medication must be returned to its secure storage immediately after the dose is administered.
3.2 Protection for Pregnant and Nursing Individuals
Specific precautions are required for household members who are pregnant, planning to become pregnant, or breastfeeding. Many oncology drugs are teratogenic, meaning they can cause developmental abnormalities in a fetus.
- Dermal Absorption Risks: Active pharmaceutical ingredients can be absorbed directly through the skin. Pregnant or nursing individuals should strictly avoid any physical contact with the medication.
- Caregiver Protocols: If a primary caregiver is pregnant and must assist with medication administration, the use of chemotherapy-rated disposable gloves is mandatory.
- Secondary Exposure: Risks extend beyond the pill itself. Minute traces of “drug dust” from a chipped tablet or chemical residue on a shared spoon or countertop can pose a cumulative risk. Ensure all surfaces used for medication preparation are thoroughly cleaned with soap and water after each use.
4. Clinical Handling Protocols for Daily Administration
Proper physical handling of oral antineoplastic agents is a critical safety requirement. Adhering to standardized handling protocols prevents the accidental transfer of cytotoxic residues to yourself, other household members, and communal surfaces.
4.1 Mandatory Hand Hygiene
Comprehensive hand hygiene is required both before and after contact with oncology medications.
- Pre-Administration: Washing with soap and warm water removes natural skin oils and contaminants that could potentially degrade the medication or interfere with its absorption.
- Post-Administration: It is essential to remove any trace chemical residues from the fingertips. Failure to wash hands after handling a tablet can lead to accidental self-exposure via the ocular or oral mucosa, or the transfer of hazardous chemicals to shared surfaces (e.g., door handles, faucets, or remote controls).
4.2 Preserving Tablet Integrity: The “Do Not Crush” Rule
Unless explicitly instructed by a prescribing oncologist or clinical pharmacist, oral oncology medications must be swallowed whole.
- Mechanism Interference: Many oncology drugs utilize enteric coatings or extended-release (ER/XR) matrices. These are engineered to ensure the drug survives the acidic environment of the stomach or is released slowly over several hours. Crushing, breaking, or chewing these tablets destroys the delivery mechanism, which can result in “dose dumping”—releasing the entire dose into the bloodstream at once, significantly increasing the risk of acute toxicity.
- Aerosolization and Drug Dust: Physically altering a tablet creates “drug dust.” This fine particulate matter can be inhaled by the patient or others in the room, leading to unintended systemic exposure.
- Clinical Alternatives: If you experience dysphagia (difficulty swallowing), do not attempt to modify the pill. Consult your medical team to discuss alternative formulations, such as liquid suspensions or smaller capsule options.
4.3 The Use of Personal Protective Equipment (PPE)
For high-potency chemotherapy tablets, the use of disposable, non-powdered gloves is a standard clinical recommendation.
- Non-Permeable Barriers: Gloves act as a critical barrier against transdermal absorption.
- Disposal Protocol: Treat used gloves as bio-hazardous waste. Use a fresh pair for every administration. After use, do not dispose of them in a standard open waste bin; place them in a dedicated, sealed plastic bag to prevent secondary exposure during household trash handling.
5. Travel Logistics: Maintaining Therapeutic Continuity
Traveling while undergoing oncology treatment requires meticulous logistical planning to ensure medication stability and regulatory compliance. Whether traveling by land or air, the primary goal is to prevent environmental excursions that could compromise the drug’s efficacy.
5.1 Environmental Security During Transit
Maintaining a stable environment is significantly more challenging during travel. Adhering to these clinical transport protocols is essential:
- Hand Luggage Requirements: Never place oncology medications in checked baggage. Aircraft cargo holds are subject to extreme atmospheric pressure changes and temperature fluctuations that can exceed the drug’s stability limits. Always keep your medication in your carry-on luggage to ensure it remains in a climate-controlled cabin environment.
- Vehicle Safety: Do not leave medications in a parked vehicle. Internal car temperatures can rapidly exceed 60°C (140°F) in direct sunlight, causing immediate chemical degradation.
- Cold-Chain Transport: For medications requiring refrigeration, utilize a high-quality medical-grade cooler bag equipped with specialized gel packs. Ensure the medication does not come into direct contact with the ice packs to prevent accidental freezing, which can permanently alter the molecular structure of biologics and certain small-molecule suspensions.
5.2 Regulatory and Emergency Documentation
Navigating airport security (TSA/International Customs) and preparing for potential emergencies requires specific medical documentation.
- Required Paperwork: Always carry a physically printed copy of your current prescription and an official “Letter of Medical Necessity” from your oncologist. This letter should explicitly state the medication’s name, the requirement for it to remain in your possession, and any necessary cooling apparatus.
- Emergency Preparedness: In the event of lost or compromised medication, having a document that lists the Generic Name, Dosage Strength, and Manufacturer is critical. This information allows local hospital pharmacies to verify and facilitate an emergency replacement of the exact formulation, minimizing gaps in your treatment cycle.
6. Environmental Stewardship: Validated Protocols for Pharmaceutical Disposal
Oral oncology medications are clinically classified as bio-active antineoplastic agents. Unlike standard pharmaceuticals, these compounds and their metabolites are highly recalcitrant—meaning they resist degradation during conventional wastewater treatment processes. Improper disposal poses a significant ecotoxicological risk, as these drugs can persist in the aquatic environment, potentially leading to bioaccumulation and endocrine disruption in wildlife.
To mitigate these risks, patients must strictly adhere to specialized disposal protocols rather than utilizing standard household waste streams.
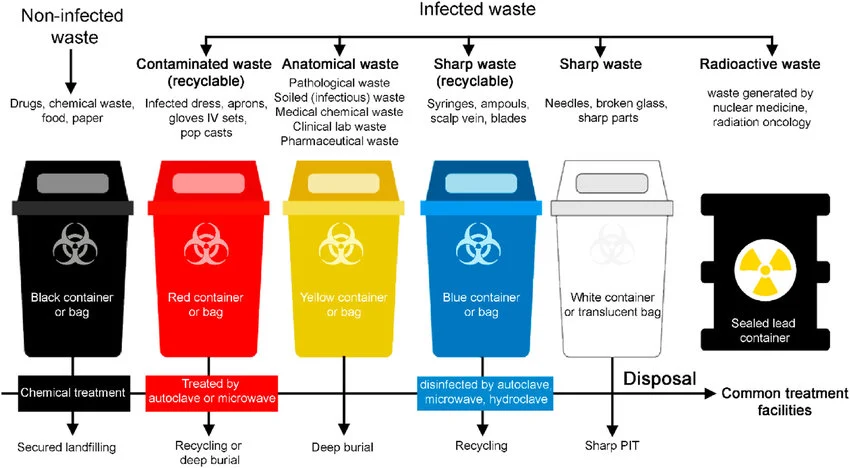
6.1 The Prohibition of Aqueous Disposal (No Flushing)
Current environmental safety regulations, including the EPA’s Hazardous Waste Pharmaceuticals Rule (Subpart P), strictly prohibit the disposal of hazardous pharmaceuticals via the sewage system.
- The Clinical Rationale: Most municipal wastewater treatment plants (WWTPs) utilize biological processes that are ineffective at neutralizing the complex molecular structures of chemotherapy drugs.
- Environmental Impact: Flushing unused tablets or pouring liquid formulations down the drain allows these cytotoxic chemicals to enter the water table in sub-therapeutic concentrations, which can harm aquatic ecosystems and potentially contaminate local drinking water sources.
6.2 Validated Disposal: High-Temperature Incineration
The only clinically approved method for the total neutralization of antineoplastic waste is high-temperature incineration at specialized, regulated facilities. This process subjects the chemical compounds to extreme heat, effectively breaking down the molecular bonds and rendering the waste inert.
6.3 Actionable Steps for Safe Disposal
If your treatment plan changes or you have expired medication, do not place it in the household trash where it could be accessed by scavengers or pets. Instead, utilize one of the following “Authorized Collection” methods:
- Pharmacy and Hospital “Take-Back” Bins: Most specialized oncology pharmacies and comprehensive cancer centers provide secure, on-site disposal kiosks. These bins are managed by stewardship organizations (such as MED-Project) and ensure the waste is transported directly to a medical waste incinerator.
- Pre-Paid Mail-Back Programs: If you are unable to visit a physical site, many manufacturers and state programs provide pre-paid, puncture-resistant mail-back envelopes. These are designed specifically for the safe transport of hazardous pharmaceuticals.
- Oncology Center Coordination: Always consult your oncology nurse or clinical pharmacist before disposing of unused pills. They can provide you with the most current list of authorized disposal sites in your immediate vicinity.
7. Medication Adherence: Leveraging Technology and Organization for Optimal Outcomes
The therapeutic success of oral oncology treatment is strictly dependent on medication adherence—taking the precise dose at the mandated clinical intervals. Maintaining a steady-state concentration of the drug in your bloodstream is essential for suppressing malignant cell growth. Sub-optimal adherence, including missed or delayed doses, can lead to therapeutic failure or the development of drug resistance.
To ensure consistency, patients should utilize a combination of physical organization and digital health interventions.
7.1 The Risks of Secondary Container Repackaging (Pill Organizers)
While multi-compartment pill organizers are widely used for chronic conditions, they are frequently contraindicated for oral oncolytics.
- Integrity Concerns: As established in Section 2, many oncology medications are highly sensitive to moisture and light. Removing a tablet from its specialized, moisture-resistant blister pack and placing it in a non-hermetic plastic organizer can lead to rapid chemical degradation.
- Cross-Contamination: Using a shared pill organizer can leave cytotoxic residues in the compartments, posing a risk to others if the organizer is subsequently used for standard vitamins or medications.
- The Clinical Gold Standard: Whenever possible, maintain the medication in its primary manufacturer packaging until the moment of administration. To track doses, use a permanent marker to record the date and time of administration directly on the exterior of the blister card or original carton.
7.2 Utilizing Digital Health Interventions (DHI)
Modern mobile health (mHealth) tools significantly improve adherence rates and facilitate more accurate communication with your clinical team.
- Automated Alerts: Utilize smartphone applications specifically designed for medication management. These platforms provide redundant alerts and “snooze” functions to ensure a dose is not overlooked during daily routines.
- Integrated Symptom Tracking: Advanced adherence applications allow patients to log side effects (such as nausea, fatigue, or dermatological changes) in real-time. This creates a high-fidelity data log for your oncologist, enabling them to make evidence-based adjustments to your supportive care plan or dosage during your next consultation.
- The Backup Protocol: Digital tools should be supplemented by a physical medication log or a “Smart Cap” system if provided by your pharmacy. These caps electronically record every time a bottle is opened, providing an objective record of your adherence history.
Clinical Adherence Tip: The “Visual Confirmation” Method “If your medication is dispensed in a bottle rather than a blister pack, do not rely on memory. Turn the bottle upside down after your morning dose and right-side up after your evening dose. This simple, tactile change provides an immediate visual confirmation of your daily adherence without exposing the medication to the environment.”
8. Acute Response Protocols: Managing Spills and Accidental Exposure
Even under the most diligent care, accidental spills or exposures can occur. Because oral oncology agents are clinically classified as cytotoxic hazardous materials, they require specialized decontamination procedures. Immediate and correct action is necessary to prevent the aerosolization of drug particles and to minimize the risk of secondary exposure to other household members.
8.1 Decontamination of Broken or Crushed Tablets
If a tablet is dropped and its integrity is compromised (chipped, crushed, or broken), the primary risk is the inhalation of drug dust. Adhere to the following clinical decontamination steps:
- Restrict Access: Immediately clear the area of children and pets. Do not allow anyone to walk through the spill zone, as this can track hazardous particles throughout the home.
- Utilize PPE: Do not attempt to clean the area with bare hands. Don a pair of disposable, non-powdered gloves (and a face mask if the tablet has been significantly crushed into powder).
- The Wet-Mopping Technique: Never use a vacuum cleaner or a dry brush to clean a broken oncology pill; this will aerosolize the hazardous dust, making it breathable. Instead, use a damp paper towel to gently pick up the fragments and wipe the surface thoroughly.
- Hazardous Waste Containment: Place the fragments, the used paper towels, and the contaminated gloves into a plastic bag. Seal this bag, then place it inside a second sealable bag (double-bagging). Label the bag clearly as “Hazardous Waste” and return it to your oncology pharmacy for high-temperature incineration.
- Surface Neutralization: After the visible debris is removed, clean the surface twice with a strong detergent or soap and water to ensure total chemical neutralization.
8.2 Emergency Protocol for Accidental Ingestion
Accidental ingestion of an antineoplastic agent by a non-patient is a medical emergency requiring immediate clinical intervention.
- Immediate Contact: Contact your local Poison Control Center or emergency services (911/local emergency number) immediately. Do not wait for symptoms (such as nausea or dizziness) to manifest; the toxic effects of oncology drugs can be delayed but severe.
- Pharmacological Identification: Have the original medication bottle or blister pack in hand. You must provide the emergency team with the exact generic name, the dosage strength (mg), and the estimated time of ingestion.
- Notify the Oncology Team: Once emergency services are contacted, notify your prescribing oncologist. They can provide the emergency room with specific clinical data regarding the drug’s half-life and potential antidotes or stabilization protocols.
9. Clinical Stewardship: The Caregiver’s Role in Medication Safety
In a home-based oncology setting, the caregiver functions as a critical clinical steward. Their role extends beyond emotional support; they are responsible for environmental compliance and the rigorous oversight of safety protocols. Data consistently indicates that patients with an active, informed caregiver experience significantly fewer medication errors and improved adherence outcomes.
By maintaining high standards for storage and handling, the caregiver ensures the patient’s treatment remains chemically stable and the household remains free from hazardous exposure.
The Weekly Medication Safety Audit
To maintain a secure environment, caregivers should perform a structured weekly safety audit. This routine check ensures that small environmental changes do not compromise the treatment plan.
- Environmental Monitoring: Verify that the storage area remains within the “Controlled Room Temperature” range (20°C–25°C). Check for new heat sources, such as seasonal sunlight shifts through windows or heat-emitting electronics placed near the medication.
- Accessibility Review: Ensure that the “high and locked” storage protocol is strictly maintained. Confirm that no medication strips or bottles have been inadvertently left on counters, bedside tables, or in common areas accessible to children or pets.
- Inventory and Expiration Check: Monitor the remaining supply to prevent gaps in the treatment cycle. Check the expiration dates on all current blister packs or bottles. If a treatment plan has changed, ensure that old or discontinued medications are immediately moved to a “disposal bag” to prevent accidental administration of the wrong drug.
- Adherence Verification: Review the medication log or adherence app with the patient. Identifying missed doses early allows the clinical team to adjust the supportive care plan before therapeutic efficacy is impacted.
Protecting the Caregiver
Vigilance is equally important for the caregiver’s own health. Caregivers must adhere to the same PPE (Personal Protective Equipment) and hand-hygiene standards as the patient. By treating every interaction with the medication as a clinical procedure, the caregiver prevents cumulative low-level exposure to cytotoxic chemicals.
🏥 Caregiver Stewardship Tip: The “Two-Person Verification” “Whenever possible, implement a ‘two-person check’ for daily doses. The caregiver verifies the drug name and dosage strength against the prescription while the patient prepares to take the pill. this simple clinical double-check is the most effective way to eliminate dosing errors at home.”
10. Conclusion: Safety as a Foundation for Clinical Success
Rigorous medication stewardship is a fundamental pillar of successful home-based oncology care. By strictly maintaining environmental storage standards and following established cytotoxic handling protocols, you preserve the drug’s therapeutic efficacy while ensuring the safety of your household and the environment.
At Generic Oncology, we are dedicated to providing the clinical evidence and practical guidelines necessary to manage these complex therapies with precision. Clinical safety is not a secondary precaution; it is an essential component of achieving optimal treatment outcomes. Stay informed, remain diligent in your safety routines, and maintain a proactive role in your clinical recovery.
Medical Disclaimer: This guide is for educational purposes only and does not substitute professional medical advice, diagnosis, or treatment. Storage and handling requirements vary significantly by medication; always follow the specific protocols provided by your oncologist or pharmacist. If you suspect your medication has been compromised or stored incorrectly, contact your clinical team immediately before taking your next dose. Medically Reviewed by Dr. Salma Elreedy