Sourcing Disclosure: Licensed Bangladesh Global Sourcing Specialist facilitating global access under Named Patient Regulations.
Expert-reviewed clinical data for prescription-only medicine.
JAK Inhibitors | Patient Guide to Clinical Efficacy, Safety, and Treatment Outcomes
Medically Reviewed by: Dr. Salma Elreedy, MD (Clinical Oncology & Internal Medicine)
Editorial Board: Generic Oncology Medical Review Board
Disclaimer: This educational hub is for informational purposes only and does not constitute medical advice. Always consult your board-certified Rheumatologist, Gastroenterologist, or Dermatologist regarding your specific treatment plan.
Managing a chronic autoimmune or inflammatory condition requires reliable, long-term medical management. In recent years, Janus Kinase (JAK) inhibitors have become a standard class of targeted medication for patients who do not respond to conventional treatments. Used across rheumatology, gastroenterology, and dermatology, these oral therapies work by blocking specific internal signals that cause inflammation. Because JAK inhibitors alter the immune system, understanding their safety profile, FDA warnings, and monitoring requirements is essential. This guide provides a clear, medically accurate breakdown of how these drugs work, the risks involved, and what to expect during treatment.
1.The Science and Mechanism
1.1 The JAK-STAT Signaling Cascade
To understand how JAK inhibitors work, it helps to visualize your immune system’s communication network as a highly coordinated chain reaction.
- The Trigger (Cytokines): Inflammatory proteins in your body called cytokines act as the initial messengers, binding to receptors on the outside surface of your cells.
- The Receptors (JAKs): This binding activates a family of enzymes attached to the inside of these receptors, known as Janus Kinases (JAKs).
- The Messengers (STATs): Once activated, the JAK enzymes stimulate a secondary group of proteins inside the cell called STATs.
- The Command Center: The STAT proteins travel directly into the cell’s nucleus, where they bind to your DNA and trigger the continuous production of inflammation.
In a healthy immune system, this pathway only activates to fight off a temporary infection. However, in autoimmune diseases, this signaling cascade is chronically overactive. Your body continuously receives the command to attack its own tissues.
The Mechanism of Action: JAK inhibitors intervene at the receptor level. By binding to and blocking the JAK enzymes, they disrupt the communication chain. The inflammatory command is intercepted before it can reach the nucleus, effectively halting the disease process at its source.
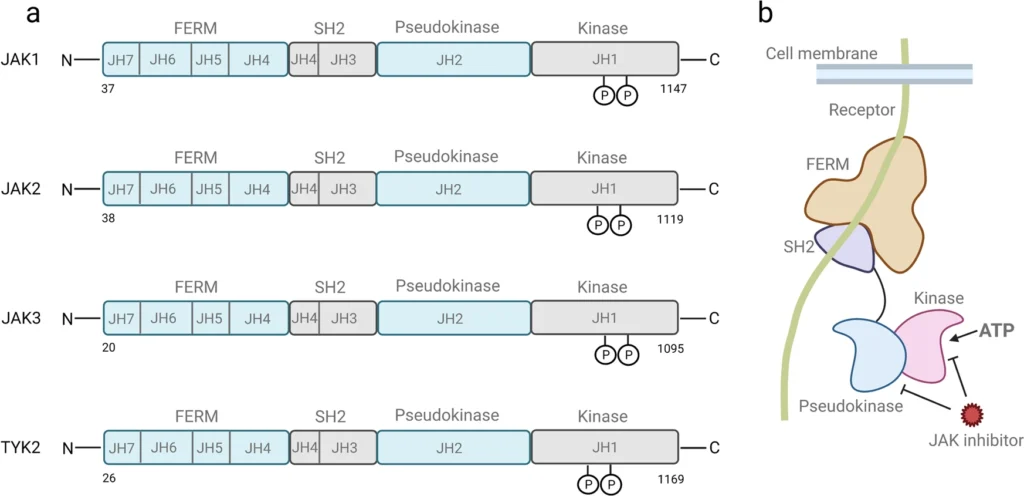
1.2 First-Generation vs. Second-Generation: The Role of Selectivity
The clinical safety and efficacy of a JAK inhibitor largely depend on its receptor selectivity.
- First-Generation (Pan-JAK Inhibitors): These initial therapies are designed to bind to and inhibit multiple JAK pathways simultaneously (typically JAK1, JAK2, and JAK3). While highly effective at suppressing systemic inflammation, this broad inhibition can also suppress normal, healthy immune functions. This wider disruption increases the potential for off-target side effects, such as viral reactivation or changes in blood counts.
- Second-Generation (Selective JAK Inhibitors): Newer therapies have been engineered to bind selectively to a single receptor subtype, such as JAK1 or TYK2. By targeting only the specific immunological pathways driving your disease, selective inhibitors aim to maintain high clinical efficacy while minimizing interference with healthy metabolic and immune functions, thereby improving the drug’s overall safety profile.
2. FDA-Approved Indications: Conditions Treated by JAK Inhibitors
JAK inhibitors have proven to be remarkably versatile, earning FDA approvals across multiple medical specialties.
| Drug Name (Generic) | Brand Name(s) | Mechanism | Major Medical Specialties & Indications |
| Tofacitini | Xeljanz | Pan-JAK | Rheumatology: Rheumatoid Arthritis (RA), Psoriatic Arthritis (PsA), Ankylosing Spondylitis. Gastroenterology: Ulcerative Colitis (UC). |
| Baricitinib | Olumiant | JAK1/JAK2 | Rheumatology: RA. Dermatology: Severe Alopecia Areata. (Also used in severe COVID-19). |
| Upadacitinib | Rinvoq | Selective JAK1 | Rheumatology: RA, PsA, Ankylosing Spondylitis. Gastroenterology: UC, Crohn’s Disease. Dermatology: Atopic Dermatitis (Eczema). |
| Ruxolitinib | Jakafi, Opzelura | JAK1/JAK2 | Hematology (Oral): Myelofibrosis, Polycythemia Vera. Dermatology (Topical): Vitiligo, Mild-to-Moderate Eczema. |
| Deucravacitinib | Sotyktu | TYK2 | Dermatology: Moderate-to-severe Plaque Psoriasis. |
3. Initiating Treatment: Clinical Pre-Screening and Safety Protocols
As targeted immunomodulators, JAK inhibitors require a rigorous clinical safety clearance prior to treatment. To mitigate potential risks, prescribing specialists strictly adhere to pre-treatment screening guidelines established by the FDA and the American College of Rheumatology (ACR). These baseline evaluations are a mandatory step to ensure the therapy is safe for your specific metabolic and immunological profile.
3.1 Required Baseline Evaluations (Pre-Screening)
- Tuberculosis (TB) Screening: A blood test (such as the QuantiFERON-TB Gold) or tuberculin skin test is mandatory. Because JAK inhibitors suppress specific immune pathways, a latent (dormant) TB infection can reactivate and become a severe illness, even if you currently exhibit no respiratory symptoms.
- Hepatitis B and C Panels: Comprehensive viral hepatitis screening is required. Pharmacological immune suppression carries a documented risk of triggering viral reactivation in patients with chronic or prior exposure to these viruses.
- Comprehensive Blood Work: Your physician will order a Complete Blood Count (CBC) to rule out pre-existing hematological issues, such as anemia or neutropenia (abnormally low white blood cells). This is paired with a Hepatic Function Panel to establish your baseline liver enzyme levels (ALT and AST).
- Baseline Lipid Panel: JAK inhibitors are known to influence lipid metabolism. A fasting cholesterol test is necessary to document your baseline LDL, HDL, and triglyceride levels before therapy begins.
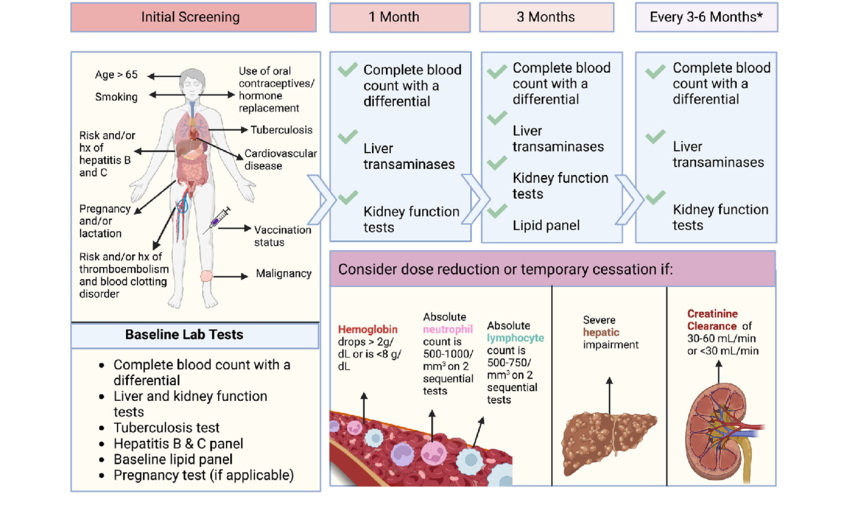
3.2 Long-Term Clinical Monitoring Protocols
Once therapy is initiated, standard clinical practice dictates routine laboratory monitoring—typically checked 4 to 8 weeks after starting the medication, and then every 3 to 6 months thereafter:
- Hepatic Function (Liver Enzymes): To continuously monitor for potential drug-induced liver toxicity.
- Hematologic Panels (CBC): To ensure your bone marrow continues to produce healthy, stable levels of red blood cells, white blood cells, and platelets without severe suppression.
- Routine Lipid Panels: It is common for patients to experience a dose-dependent elevation in cholesterol. If hyperlipidemia occurs, the standard clinical protocol is often to prescribe a lipid-lowering medication (such as a statin) to manage cardiovascular risk, allowing you to safely remain on the JAK inhibitor.
4. Safety, Risk Stratification, and the FDA Boxed Warning
Transparency regarding adverse events is a cornerstone of responsible medical management. In 2021, the safety profile of oral JAK inhibitors underwent intense regulatory scrutiny following the publication of the ORAL Surveillance trial in the New England Journal of Medicine. Based on these clinical findings, the FDA mandated class-wide “Boxed Warnings” (the strictest regulatory safety alert) for JAK inhibitors used to treat inflammatory conditions.
Understanding the context of these warnings and how physicians stratify patient risk is essential for making an informed treatment decision.
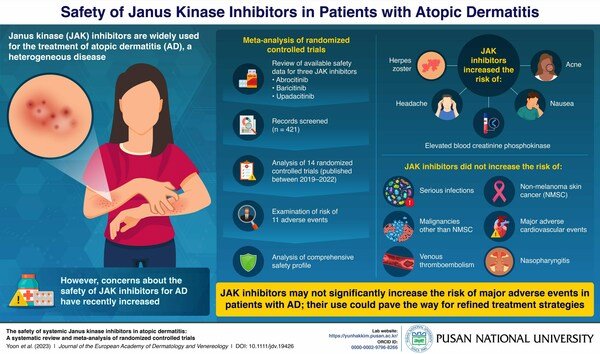
4.1 The Clinical Context: The ORAL Surveillance Trial
To accurately interpret the FDA warnings, it is critical to understand the patient population studied in the ORAL Surveillance trial. The study specifically evaluated patients taking tofacitinib (Xeljanz) who were over the age of 50 and had at least one pre-existing cardiovascular risk factor. In this specific, older, high-risk demographic, researchers observed slightly higher incidences of cardiovascular and malignancy events compared to patients taking TNF-alpha inhibitors (like adalimumab or etanercept). The FDA subsequently applied these warnings to the entire class of systemic JAK inhibitors.
4.2 Decoding the FDA Boxed Warnings
The mandated warnings alert physicians and patients to four primary categories of severe, though statistically uncommon, adverse events:
- MACE (Major Adverse Cardiovascular Events): This category includes myocardial infarction (heart attack), stroke, and cardiovascular-related death. The data indicates an elevated hazard ratio for MACE in patients treated with JAK inhibitors compared to those on TNF blockers, particularly among patients with an extensive smoking history.
- VTE (Venous Thromboembolism): There is an increased risk of blood clots, specifically Deep Vein Thrombosis (DVT) in the extremities and Pulmonary Embolism (PE) in the lungs. Clinical data suggests this risk may be dose-dependent in certain JAK inhibitors.
- Malignancy: The immunosuppressive nature of these therapies carries a slightly elevated risk for developing certain cancers. The most frequently observed in clinical trials are Non-Melanoma Skin Cancers (NMSC), such as basal or squamous cell carcinomas, and, less commonly, lymphomas.
- Serious Opportunistic Infections: By intentionally dampening the immune system’s inflammatory pathways, JAK inhibitors reduce the body’s defense against pathogens. This can lead to severe bacterial, mycobacterial, invasive fungal, or viral infections that may require hospitalization.
4.3 Clinical Risk Stratification: High-Risk vs. Low-Risk Profiles
In modern clinical practice, rheumatologists and dermatologists use risk stratification protocols. The absolute risk of a severe adverse event depends heavily on the patient’s baseline health.
- The High-Risk Cohort: The FDA advises against using JAK inhibitors as a first-line therapy for patients who are over the age of 65, possess a history of Atherosclerotic Cardiovascular Disease (ASCVD), have a known hypercoagulable state (prone to blood clots), or have a current or past history of chronic smoking. For these patients, specialists typically exhaust conventional DMARDs and biologic therapies before considering a JAK inhibitor.
- The Low-Risk Cohort: For younger patients (under 50) without a smoking history, normal lipid profiles, and no cardiovascular comorbidities, the absolute risk of MACE or VTE remains exceedingly low. In these cases, the clinical consensus often determines that the benefits of halting severe, tissue-damaging inflammation heavily outweigh the statistical risks.
4.4 Common Adverse Events and Day-to-Day Management
While severe complications are rare, day-to-day side effects are more common and require standard clinical management:
- Herpes Zoster (Shingles) Reactivation: JAK inhibitors specifically suppress the interferon pathways that keep dormant viruses in check. Consequently, there is a well-documented increased risk of Varicella-Zoster Virus (VZV) reactivation. Clinical Protocol: The American College of Rheumatology strongly recommends that all patients receive the Recombinant Zoster Vaccine (Shingrix) prior to initiating JAK inhibitor therapy, regardless of age.
- Dermatological Events: Selective JAK1 inhibitors (such as upadacitinib) are frequently associated with drug-induced follicular acne. This is usually mild to moderate and can be managed effectively with topical dermatological treatments without discontinuing the JAK inhibitor.
- Upper Respiratory Tract Infections (URTIs): Nasopharyngitis (the common cold), sinus infections, and mild bronchitis are the most frequently reported adverse events across all clinical trials for this drug class.
- Headaches and Nausea: Mild, transient headaches and gastrointestinal upset may occur during the initial weeks of treatment as the body acclimates to the medication.
5. Future Innovations: Pipeline Indications and Advanced Delivery Mechanisms
The clinical landscape of Janus Kinase inhibition is advancing rapidly beyond its initial indications. As researchers map the precise cytokine pathways responsible for various immune-mediated diseases, the next decade of JAK therapy will be defined by novel delivery systems and expanded clinical applications, including breakthrough research in endocrinology and transplant oncology.
5.1 Topical Formulations: Maximizing Efficacy While Sparing Systemic Exposure
One of the most significant pharmacological advancements is the successful formulation of topical JAK inhibitors. While oral therapies distribute systemically via the bloodstream—necessitating the FDA boxed warnings for cardiovascular and hematological risks—topical therapies are engineered to bypass systemic circulation.
- Pharmacokinetics of Topical Ruxolitinib (Opzelura): FDA-approved for mild-to-moderate atopic dermatitis and non-segmental vitiligo, topical ruxolitinib (1.5% cream) penetrates the stratum corneum to deliver concentrated localized immunosuppression directly to the affected dermal layers.
- The Safety Advantage: Clinical pharmacokinetic data demonstrates that topical application results in steady-state plasma concentrations that are drastically lower than those seen with oral dosing. By keeping the drug localized to the skin, patients achieve high clinical efficacy while effectively eliminating the risk of systemic adverse events (such as VTE or MACE).
- Dermatological Horizons: Researchers are currently expanding clinical trials for topical JAKs to treat a wider array of localized, treatment-resistant inflammatory dermatoses—including severe localized psoriasis and chronic seborrheic conditions—where long-term use of high-potency topical corticosteroids is contraindicated due to skin atrophy.
5.2 Emerging Clinical Horizons: The Therapeutic Pipeline
Because the JAK-STAT pathway is central to so many cellular communication networks, clinical trials are investigating the efficacy of these small molecules across a diverse spectrum of complex systemic diseases:
- Endocrinology (Type 1 Diabetes): Groundbreaking phase 2 clinical trials (recently highlighted in the New England Journal of Medicine) have investigated the use of oral baricitinib in newly diagnosed Type 1 Diabetes patients. The data suggests that JAK1/JAK2 inhibition can suppress the autoimmune destruction of pancreatic beta cells, successfully preserving the body’s natural insulin production (measured by C-peptide levels) and reducing the reliance on exogenous insulin therapy.
- Systemic Lupus Erythematosus (SLE): SLE is characterized by a high “Type I Interferon signature,” a signaling pathway heavily dependent on the TYK2 enzyme. Next-generation, highly selective TYK2 inhibitors (like deucravacitinib) are currently in late-stage phase trials for lupus. By precisely targeting this pathway, rheumatologists aim to control lupus flares without the broad immunosuppression that leaves patients vulnerable to severe infections.
- Severe Hidradenitis Suppurativa (HS): HS is a chronic, severely debilitating inflammatory skin condition with very few approved systemic treatments. Upadacitinib (a selective JAK1 inhibitor) is currently showing highly promising efficacy in Phase 3 trials, drastically reducing inflammatory nodules and abscesses in patients who have failed standard biologic therapies.
- Hematopoietic Stem Cell Transplantation & Oncology: While traditionally used for autoimmune conditions, JAK inhibitors are becoming pivotal in malignant hematology and oncology care. Ruxolitinib is now an FDA-approved, critical intervention for steroid-refractory Acute and Chronic Graft-Versus-Host Disease (GVHD)—a life-threatening complication following allogeneic stem cell transplants for blood cancers. By dampening the donor T-cell response, JAK inhibitors are fundamentally improving survival outcomes for transplant patients.
6. Patient Empowerment: Navigating Treatment Through Shared Decision-Making
Modern clinical practice relies heavily on “shared decision-making”—a collaborative approach where your physician’s medical expertise is combined with your personal lifestyle, values, and risk tolerance. Because starting a systemic immunomodulator is a significant commitment, you must be an active advocate for your own health.
To bridge the gap between the exam room and your daily life, use the following framework as your personal discussion guide. Bring this checklist to your next appointment to ensure your treatment plan is tailored specifically to you.
6.1 The Clinical Discussion Guide: 5 Critical Questions for Your Prescribing Specialist
Instead of simply asking what the drug is, use these targeted questions to understand exactly how the medication will interact with your unique biology and daily routine.
Q. “Based on my complete medical and family history, how do you stratify my personal risk regarding the FDA Boxed Warnings?”
- Why you must ask this: The FDA warnings for MACE (cardiovascular events) and blood clots primarily impact specific demographics.
- What to look for in the answer: Your doctor should review your specific age, smoking history, lipid panels, and family history of cardiovascular disease or cancer before confirming whether you fall into a high-risk or low-risk cohort for this specific class of drugs.
Q. “Are all my required immunizations—specifically the Recombinant Zoster (Shingles) vaccine—up to date before we initiate immune suppression?”
- Why you must ask this: JAK inhibitors drastically increase the risk of viral reactivation, particularly for Shingles. Furthermore, once you start the medication, you generally cannot receive “live” vaccines.
- What to look for in the answer: A clear timeline. Your physician should ideally coordinate with your primary care provider to ensure you receive the Shingrix, Pneumonia, and current Influenza/COVID-19 vaccines before taking your first pill.
Q. “What is our exact ‘Sick Day Protocol’ if I develop a common infection, and at what temperature fever should I pause the medication?”
- Why you must ask this: Upper respiratory infections are the most common side effect. You need to know when a simple cold warrants stopping the drug to let your immune system fight.
- What to look for in the answer: Clear, actionable parameters. For example, a doctor might advise: “Continue the medication for a runny nose, but if you develop a productive cough, painful urination, or a fever over 100.4°F (38°C), hold the medication and call the clinic.”
Q. “What is the logistical expectation for my routine lab monitoring, and can these orders be coordinated with a local lab facility?”
- Why you must ask this: JAK inhibitors require ongoing surveillance of your liver enzymes, complete blood counts (CBC), and lipid panels.
- What to look for in the answer: A predictable schedule (e.g., “We will test at week 4, week 12, and then every 3 months”). Knowing this upfront allows you to prepare for the logistical commitment of long-term therapy.
Q. “Given my specific diagnosis, is a second-generation (selective) JAK inhibitor an option, or does my insurance require ‘Step Therapy’ first?”
- Why you must ask this: Newer, highly selective JAK inhibitors (targeting only JAK1 or TYK2) often boast a more refined safety profile with fewer off-target side effects than older, Pan-JAK inhibitors.
- What to look for in the answer: A transparent discussion about both medical efficacy and financial realities. Sometimes, insurance companies mandate that you try and fail older or less expensive biologics before they will approve the newest selective JAK inhibitors.
6.2 The “When to Call the Clinic” Safety Checklist
Part of patient empowerment is knowing the difference between a manageable side effect and an urgent clinical event. Do not wait for your next appointment if you experience any of the following while on a JAK inhibitor; contact your specialist or seek urgent care immediately:
- Signs of a Blood Clot (VTE/DVT): Unexplained, sudden swelling, warmth, or deep pain in one leg or arm.
- Signs of a Pulmonary Embolism (PE): Sudden shortness of breath, sharp chest pain that worsens when you take a deep breath, or coughing up blood.
- Signs of Herpes Zoster (Shingles): A painful, burning, or tingling blistering rash, especially if it appears in a stripe or cluster on one side of your face or torso.
- Signs of Severe Infection: A sustained fever, severe localized pain, or a skin lesion that becomes rapidly red, hot, and swollen.
7. Real-World Patient Case Studies: Clinical Outcomes in Ulcerative Colitis
The following case studies are synthesized from documented patient accounts on gastroenterology support forums, illustrating both the initial devastation of an uncontrolled flare and the long-term success of transitioning to a JAK inhibitor.
7.1 Case Study 1: Overcoming Biologic Immunogenicity with a JAK Inhibitor (Xeljanz)
The Clinical Profile: A patient with a 6-year documented history of Ulcerative Colitis, sharing their treatment progression. The Baseline Presentation: The patient initially achieved clinical remission using an intravenous anti-TNF biologic (infliximab/Remicade), which remained highly effective for over four years. However, the patient eventually experienced a secondary loss of response. Clinical evaluation determined that the patient’s immune system had developed anti-drug antibodies against the large biologic proteins, rendering the medication ineffective and causing a resurgence of disease activity. The patient noted significant psychological distress and fear regarding running out of viable treatment options. The Therapeutic Intervention: To bypass the issue of immunogenicity, the treating gastroenterologist transitioned the patient from the large-molecule biologic to an oral small-molecule Pan-JAK inhibitor (tofacitinib/Xeljanz), prescribed at 10mg twice daily. The Clinical Outcome: The patient successfully recaptured disease control. At the time of their report, they had maintained solid, sustained clinical remission for two consecutive years on the oral JAK inhibitor therapy. Clinical Takeaway: This case highlights a primary advantage of JAK inhibitors: because they are small-molecule therapies, the body does not form antibodies against them, making them a highly durable second-line option for patients who fail TNF-blockers.
7.2 Case Study 2: The Baseline Impact of Uncontrolled Inflammation
(Note: While this patient is currently on early-stage therapy, their account perfectly illustrates the severe baseline conditions that frequently necessitate rapid escalation to advanced therapies like JAK inhibitors.) The Clinical Profile: A young adult male recently diagnosed with left-sided Ulcerative Colitis. The Baseline Presentation: The patient presented with an acute, severe initial flare. Reported symptoms included chronic, daily hematochezia (bloody stools), severe gastrointestinal distress, and the inability to retain food. Systemically, the patient experienced rapid, unintended weight loss and a severe reduction in muscle mass. The Functional Impact: The physical toll of the uncontrolled inflammation severely impaired the patient’s daily functioning. He reported being unable to participate in regular physical activities (sports) and documented a significant decline in professional performance (missing sales targets) due to the frequency and urgency of his symptoms, compounded by the physical exhaustion of undergoing diagnostic procedures like a colonoscopy. The Therapeutic Intervention & Prognosis: The patient was initially prescribed a first-line aminosalicylate (mesalamine). Clinical Takeaway: This raw account underscores the profound physiological and psychological devastation of active Inflammatory Bowel Disease. If this patient fails to achieve mucosal healing on standard first-line treatments like mesalamine, his presentation makes him a prime candidate for rapid escalation to targeted therapies—such as selective JAK inhibitors—to quickly halt the inflammatory cascade, prevent permanent bowel damage, and restore his professional and personal quality of life.
7.3 The “When to Call the Clinic” Safety Checklist
Part of patient empowerment is knowing the difference between a manageable side effect and an urgent clinical event. Do not wait for your next appointment if you experience any of the following while on a JAK inhibitor; contact your specialist or seek urgent care immediately:
- Signs of a Blood Clot (VTE/DVT): Unexplained, sudden swelling, warmth, or deep pain in one leg or arm.
- Signs of a Pulmonary Embolism (PE): Sudden shortness of breath, sharp chest pain that worsens when you take a deep breath, or coughing up blood.
- Signs of Herpes Zoster (Shingles): A painful, burning, or tingling blistering rash, especially if it appears in a stripe or cluster on one side of your face or torso.
- Signs of Severe Infection: A sustained fever, severe localized pain, or a skin lesion that becomes rapidly red, hot, and swollen.
8. Pharmacological Profiles and Clinical Evidence: The Major JAK Inhibitors
Therapeutic efficacy in the Janus Kinase inhibitor class is established through rigorous, multi-center Phase 3 clinical trial programs. Because each medication possesses a unique receptor binding profile, understanding their distinct mechanisms and the clinical data that secured their FDA approvals is essential for evidence-based treatment selection.
8.1 Tofacitinib (Brand Name: Xeljanz)
- Manufacturer: Pfizer
- FDA-Approved Indications: Rheumatoid Arthritis (RA), Psoriatic Arthritis (PsA), Ulcerative Colitis (UC), Ankylosing Spondylitis (AS), and Polyarticular Course Juvenile Idiopathic Arthritis (pcJIA).
- Mechanism of Action: A first-generation, pan-JAK inhibitor with preferential affinity for JAK1 and JAK3. By inhibiting these specific pathways, tofacitinib effectively blunts the intracellular signaling of multiple pro-inflammatory interleukins (including IL-2, IL-4, IL-6, IL-15, and IL-21) responsible for immune dysregulation.
8.1.1 The Clinical Evidence: ORAL and OCTAVE Trial Programs
As the first oral JAK inhibitor introduced to the clinical landscape, tofacitinib’s efficacy is supported by extensive long-term data.
- In Rheumatology (The ORAL Trials): Across thousands of patients with moderate-to-severe RA, tofacitinib demonstrated statistically significant efficacy. At month 3, approximately 59% of patients achieved an ACR20 response (a standard 20% improvement in tender and swollen joint counts alongside other clinical measures), compared to 25% on placebo. Furthermore, radiological assessments at month 6 confirmed that 73% of patients experienced no radiographic progression, proving the drug actively halts structural joint damage.
- In Gastroenterology (The OCTAVE Trials): The OCTAVE Induction 1, 2, and Sustain trials evaluated patients with moderate-to-severe UC who had previously failed TNF blockers or corticosteroids. Tofacitinib demonstrated a rapid onset of action, with patients noting a significant reduction in rectal bleeding and stool frequency within days. By week 8 of the 10mg induction dose, 16.6% to 18.5% of patients achieved stringent clinical remission (compared to 3.6% to 8.2% on placebo). During the 52-week maintenance phase, over 40% of patients successfully sustained clinical remission and mucosal healing.
8.2 Upadacitinib (Brand Name: Rinvoq)
- Manufacturer: AbbVie
- FDA-Approved Indications: Rheumatoid Arthritis, Psoriatic Arthritis, Atopic Dermatitis, Ulcerative Colitis, Crohn’s Disease, Ankylosing Spondylitis, and Non-radiographic Axial Spondyloarthritis.
- Mechanism of Action: A second-generation, reversible, and highly selective JAK1 inhibitor. Upadacitinib was specifically engineered to maximize anti-inflammatory efficacy while theoretically minimizing the JAK2 and JAK3-associated off-target hematological and immunological effects seen in older therapies.
8.2.1 The Clinical Evidence: SELECT and U-ACHIEVE Trial Programs
- In Rheumatology (The SELECT Trials): Upadacitinib achieved clinical superiority in highly competitive head-to-head trials. In the phase 3 SELECT-COMPARE study evaluating RA patients on background methotrexate, upadacitinib was tested directly against adalimumab (Humira). The results established clinical superiority: 43.3% of patients taking upadacitinib achieved low disease activity by week 12, compared to 22.4% on adalimumab.
- In Gastroenterology (The U-ACHIEVE and U-ACCOMPLISH Trials): In the treatment of steroid-refractory Ulcerative Colitis, the phase 3 data established upadacitinib as a highly potent intervention. At week 8 of the 45mg induction phase, 33% of patients achieved clinical remission compared to 4% on placebo. More critically, endoscopic evaluations confirmed profound mucosal healing, proving the medication actively repairs the intestinal lining rather than merely masking systemic symptoms.
8.3 Baricitinib (Brand Name: Olumiant)
- Manufacturer: Eli Lilly
- FDA-Approved Indications: Rheumatoid Arthritis, Severe Alopecia Areata, and severe COVID-19 (in hospitalized adults requiring supplemental oxygen).
- Mechanism of Action: A reversible, selective JAK1 and JAK2 inhibitor. By modulating the JAK1/JAK2 pathways, it effectively disrupts the signaling of inflammatory cytokines heavily implicated in both joint destruction and hair follicle autoimmunity.
8.3.1 The Clinical Evidence: RA-BEAM and BRAVE-AA Trial Programs
- In Rheumatology (The RA-BEAM Trial): Baricitinib demonstrated robust efficacy in the RA-BEAM study, establishing statistical superiority over both placebo and adalimumab (Humira) in reducing RA disease activity and rapidly alleviating the duration of morning joint stiffness.
- In Dermatology (The BRAVE-AA Trials): Baricitinib represents a paradigm shift in dermatological care, becoming the first FDA-approved systemic therapy for severe Alopecia Areata. The phase 3 BRAVE-AA1 and BRAVE-AA2 trials evaluated 1,200 adults with a baseline Severity of Alopecia Tool (SALT) score indicating at least 50% scalp hair loss. The results were historic: approximately 35% to 39% of patients taking the 4mg daily dose achieved at least 80% scalp hair coverage by week 36. Furthermore, roughly 1 in 3 patients achieved full regrowth or minimal gaps in their eyebrow and eyelash hair, proving that targeted JAK inhibition can successfully reverse the autoimmune attack on dormant hair follicles.
8.4 Ruxolitinib 1.5% Cream (Brand Name: Opzelura)
- Manufacturer: Incyte
- FDA-Approved Indications: Short-term and non-continuous chronic treatment of mild to moderate Atopic Dermatitis (in non-immunocompromised patients aged 12 and older); Non-segmental Vitiligo (in patients aged 12 and older).
- Mechanism of Action: A potent, selective JAK1 and JAK2 inhibitor formulated specifically for topical administration. By applying the therapy directly to the affected epidermis, ruxolitinib locally blunts the cytokine signaling pathways (including interferon-gamma and specific interleukins) responsible for cutaneous inflammation and autoimmune melanocyte destruction. Critically, this topical formulation minimizes systemic absorption, significantly mitigating the off-target hematological and cardiovascular risks associated with oral JAK inhibitors.
8.4.1 The Clinical Evidence: TRuE-AD and TRuE-V Trial Programs
As the first topical JAK inhibitor approved by the FDA, ruxolitinib cream represents a major advancement in dermatological pharmacology, allowing for localized immunosuppression without the need for systemic exposure.
- In Atopic Dermatitis (The TRuE-AD Trials): The phase 3 TRuE-AD1 and TRuE-AD2 clinical trials evaluated over 1,200 patients with mild-to-moderate eczema. The primary clinical endpoint was Investigator’s Global Assessment (IGA) success—defined as clear or almost clear skin with at least a 2-grade improvement from baseline. At week 8, over 50% of patients applying the 1.5% ruxolitinib cream achieved IGA success, compared to approximately 15% applying a non-medicated vehicle cream. Furthermore, patients reported a rapid, statistically significant reduction in pruritus (severe itching), with measurable relief often recorded within 12 hours of the initial application.
- In Vitiligo (The TRuE-V Trials): Opzelura achieved a historic clinical milestone by becoming the first FDA-approved medical treatment designed to induce follicular repigmentation in non-segmental vitiligo. The phase 3 TRuE-V1 and TRuE-V2 trials utilized the Facial Vitiligo Area Scoring Index (F-VASI) as a primary metric. At week 24 of continuous application, approximately 30% of patients achieved an F-VASI75 response (a 75% or greater improvement in facial repigmentation) compared to roughly 10% on the vehicle cream. During the 52-week open-label extension period, clinical efficacy continued to compound; approximately 50% of patients ultimately achieved the F-VASI75 endpoint, proving that localized JAK inhibition can effectively halt and reverse the autoimmune destruction of pigment-producing cells over time.
9. People Also Ask
How rapidly do JAK inhibitors take effect?
Because they are small molecules that rapidly penetrate cells, onset of action is swift. Patients often observe a clinical response (reduction in joint swelling or bowel urgency) within 1 to 2 weeks, with peak therapeutic efficacy generally achieved between 3 and 6 months of continuous therapy.
Is strict adherence to a daily dosing schedule required?
Are there dietary restrictions for administration?
What is the protocol for a missed dose?
Does therapeutic efficacy wane over time (immunogenicity)?
Are JAK inhibitors classified as chemotherapy?
Why do selective JAK inhibitors frequently cause acne?
Do these medications induce weight gain?
Is alcohol consumption contraindicated?
Should therapy be withheld during active infections?
Why is the Herpes Zoster (Shingles) vaccine highly emphasized?
Does hyperlipidemia require drug discontinuation?
Is the development of Venous Thromboembolism (VTE) highly probable?
Can a JAK inhibitor be combined with a Biologic therapy?
What is the “washout” protocol when transitioning therapies?
Is body art (tattoos/piercings) safe during treatment?
How does this impact surgical procedures?
10. Critical Questions for Your Prescribing Specialist:
- Risk Stratification: “Based on my comprehensive medical history—specifically my age, lipid baseline, smoking history, and any familial history of atherosclerotic cardiovascular disease (ASCVD) or malignancy—do I fall into a high-risk or low-risk cohort regarding the FDA Boxed Warnings for this specific therapy?”
- Immunization Protocols: “Are my required vaccinations current? Specifically, how should we schedule the recombinant zoster (Shingrix) and pneumococcal vaccines to ensure maximum immunological response prior to initiating baseline immunosuppression?”
- Infection Management (Sick Day Rules): “What is our exact clinical protocol for temporary drug cessation in the event of an infection? Specifically, at what body temperature or severity of respiratory symptoms should I withhold my daily dose and contact the clinic?”
- Laboratory Surveillance: “What is the required interval for routine laboratory monitoring (specifically hepatic function panels, complete blood counts, and lipid profiles), and can these diagnostic orders be coordinated with my primary care physician or a local facility?”
- Receptor Selectivity: “Given my specific primary diagnosis and comorbidity profile, is a second-generation (highly selective JAK1 or TYK2) inhibitor clinically indicated over a first-generation (Pan-JAK) option to potentially minimize off-target hematological side effects?”
The era of “one-size-fits-all” autoimmune treatment is over. Janus Kinase inhibitors represent a monumental leap forward in precision medicine, offering the convenience of an oral pill paired with the heavy-hitting efficacy previously reserved for IV infusions. By understanding the science, respecting the safety protocols, and maintaining open communication with your medical team, you can confidently utilize these therapies to reclaim your quality of life.