Sourcing Disclosure: Licensed Bangladesh Global Sourcing Specialist facilitating global access under Named Patient Regulations.
Expert-reviewed clinical data for prescription-only medicine.
Molecular Mechanisms of Synthetic Lethality: A Clinical Review of PARP Inhibition in BRCA-Mutated Malignancies
Medically Reviewed by: Dr. Salma Elreedy, Clinical Oncologist, Sphinx Cure Oncology Center.
Fact-Checked by: Generic Oncology Editorial Team.
Synthetic lethality is a genetic phenomenon where the simultaneous loss of function in two separate genes results in cell death, while the loss of either gene individually is compatible with cellular viability. In oncology, this concept has been successfully translated into clinical practice through the development of poly (ADP-ribose) polymerase (PARP) inhibitors. This therapeutic strategy exploits the pre-existing DNA repair defects in certain malignancies, most notably those harboring BRCA1 or BRCA2 mutations, to induce targeted apoptosis while sparing healthy, repair-proficient cells.
Summary for Patients and Caregivers
- Targeted Action: Olanib (Olaparib) is not a conventional chemotherapy; it is a “targeted therapy” that exploits a specific weakness in cancer cells.
- The Mechanism: Cancer cells with BRCA1 or BRCA2 mutations already have a broken DNA repair system. Olanib blocks a second repair protein called PARP, leaving the cancer cell with no way to fix itself.
- The Outcome: This “double-hit” causes the cancer cell to die while leaving healthy cells (which have at least one working repair system) relatively unaffected.
- Clinical Goal: To extend progression-free survival (PFS) in ovarian, breast, prostate, and pancreatic cancers.
1. The Clinical Rationale: Defining Synthetic Lethality
Synthetic lethality is a pharmacological strategy where the simultaneous deficiency of two genes or pathways leads to cell death, whereas a deficiency in only one is non-lethal. In oncology, this is achieved by administering a PARP inhibitor (PARPi) to patients whose tumors already harbor Homologous Recombination Deficiency (HRD)—most commonly via BRCA1 or BRCA2 mutations.
DNA Damage and the PARP Pathway
Human cells experience approximately 104 to 105 DNA lesions daily. Single-strand breaks (SSBs) are the most frequent and are primarily repaired by the Base Excision Repair (BER) pathway. PARP1 (Poly [ADP-ribose] polymerase 1) is the essential sensor for these breaks. Upon detecting a lesion, PARP1 utilizes NAD+ to synthesize PAR polymers, which recruit repair enzymes like XRCC1.
2. Mechanism of Action: Beyond Simple Inhibition
Olanib (Olaparib) exerts its anti-tumor effect through two distinct molecular processes:
- Catalytic Inhibition: The molecule binds to the NAD+ binding pocket of PARP1/2, preventing the formation of PAR chains. This halts the recruitment of the BER machinery, leaving SSBs unrepaired.
- PARP Trapping: This is the more cytotoxic mechanism. Olanib “traps” the PARP protein onto the DNA at the site of the break. These stable PARP-DNA complexes act as physical barriers that stall and collapse replication forks during the S-phase of the cell cycle.
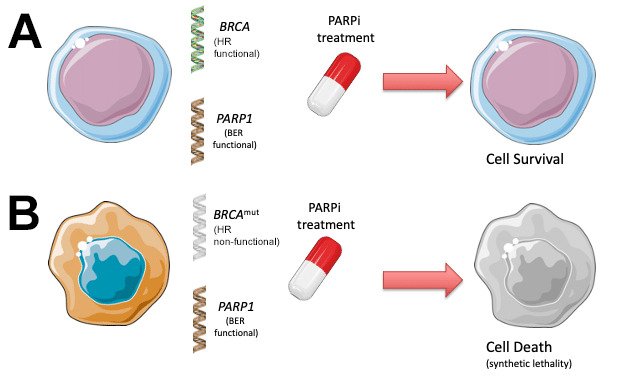
3. The Conversion to Lethal Double-Strand Breaks (DSBs)
When a replication fork collapses due to a trapped PARP complex, the SSB is converted into a Double-Strand Break (DSB).
- In Healthy Cells: The Homologous Recombination Repair (HRR) pathway (facilitated by functional BRCA proteins) repairs the DSB with high fidelity.
- In BRCA-Mutated Cells: The HRR pathway is non-functional. The cell is forced to use error-prone pathways like Non-Homologous End Joining (NHEJ), leading to catastrophic genomic instability and apoptosis (programmed cell death).
| Clinical Trial | Patient Population | Primary Endpoint | Hazard Ratio (HR) | P-Value |
| SOLO-1 | gBRCAm Advanced Ovarian Cancer | 70% reduction in risk of progression | 0.30 | < 0.001 |
| OlympiAD | gBRCAm HER2- Metastatic Breast Cancer | Median PFS 7.0 mo vs. 4.2 mo (chemo) | 0.58 | 0.0009 |
| PROfound | mCRPC with BRCA1/2 or ATM mutations | Significant improvement in rPFS | 0.34 | < 0.0001 |
4. Safety Profile and Clinical Monitoring (YMYL Requirement)
While targeted, Olanib requires proactive clinical management to mitigate hematological and gastrointestinal toxicities.
- Hematological Toxicity: Anemia is the most frequent Grade 3 or higher adverse event, occurring in approximately 18-20% of patients in the SOLO-1 and OlympiAD trials. Neutropenia and thrombocytopenia are also observed.
- Secondary Malignancies: There is a rare but documented risk (< 1.5\%) of Myelodysplastic Syndrome (MDS) or Acute Myeloid Leukemia (AML), particularly in patients previously treated with platinum-based chemotherapy.
- Monitoring Protocol: 1. Baseline: Complete Blood Count (CBC) and renal function tests.2. Ongoing: Monthly CBC for the first 12 months of treatment.3. Dose Adjustment: Persistent cytopenias may require dose interruption or reduction to 250 mg or 200 mg twice daily.
5. Patient Stratification: Identifying “BRCAness”
The efficacy of Olanib is not limited to germline mutations (gBRCAm). The concept of “BRCAness” refers to tumors that exhibit HRD due to somatic mutations, ATM loss, or BRCA1 promoter methylation. Clinical decision-making now relies on HRD Genomic Instability Scores to identify patients who are likely to respond to PARP inhibition despite lacking a germline mutation.
| Inhibitor | Catalytic Inhibition ((IC50)) | PARP Trapping Potency | Clinical Application |
| Talazoparib (Talaparib) | Highly Potent | High (100x vs Olaparib) | gBRCAm Breast Cancer |
| Niraparib (Niranib) | Potent | Moderate-High | Ovarian Cancer (Maintenance) |
| Olaparib (Olanib) | Potent | Moderate | Ovarian, Breast, Prostate, Pancreatic |
| Rucaparib | Potent | Moderate | Ovarian, Prostate |
Synthetic lethality through PARP inhibition represents a paradigm shift in precision oncology. By moving from non-specific cytotoxic agents to molecularly targeted inhibition, clinicians can exploit the inherent weaknesses of the cancer genome. Olanib (Olaparib) continues to be the foundational molecule in this space, providing a highly effective option for patients with DNA repair deficiencies across multiple solid tumor types.
Clinical Insight: Monitoring in Practice
“In a clinical setting, we often see the most significant drop in hemoglobin (anemia) within the first 3 to 8 weeks of starting Olanib. While the synthetic lethality mechanism is highly targeted, the impact on bone marrow is a known class effect of PARP inhibitors. I advise patients to report any unusual fatigue or shortness of breath immediately, as a proactive dose interruption for 1–2 weeks often allows the marrow to recover without compromising the drug’s long-term efficacy.” — Dr. Salma Elreedy
Frequently Asked Questions (FAQs)
How does Synthetic Lethality differ from standard Cytotoxic Chemotherapy?
Traditional chemotherapy primarily targets all rapidly dividing cells, which leads to significant off-target toxicity (e.g., hair loss, mucosal damage). In contrast, Synthetic Lethality is a form of “precision oncology.” It exploits a pre-existing genetic defect (the “first hit,” such as a BRCA1/2 mutation) that is only present in cancer cells. By introducing a PARP inhibitor (the “second hit”), the therapy induces cell death specifically in the tumor, sparing healthy cells that retain a functional DNA repair pathway.
Who is the ideal candidate for Olanib (Olaparib)?
Patients with advanced ovarian, breast, pancreatic, or prostate cancer who have a confirmed BRCA1 or BRCA2 mutation or other evidence of Homologous Recombination Deficiency (HRD).
How long does a patient stay on Olanib treatment?
In a maintenance setting (like after first-line chemotherapy for ovarian cancer), treatment typically continues for up to 2 years or until disease progression or unacceptable toxicity occurs.
Can Olanib be taken with other medications?
Olaparib is metabolized by the CYP3A enzyme. Patients should avoid strong CYP3A inhibitors (e.g., clarithromycin) or inducers (e.g., rifampin), and should not consume grapefruit or Seville oranges during treatment.
What is “PARP Trapping,” and why is it more important than simple inhibition?
Simple inhibition just stops the PARP enzyme from working. PARP Trapping occurs when the drug locks the PARP enzyme onto the DNA strand. These “trapped” complexes are 100 times more toxic to the cancer cell than unrepaired DNA breaks because they physically block the cell’s ability to copy its DNA, leading to immediate replication fork collapse.
Scientific References
- SOLO-1 Trial: Moore M, et al. “Maintenance Olaparib in Patients with Newly Diagnosed Advanced Ovarian Cancer.“ New England Journal of Medicine (2018). [DOI: 10.1056/NEJMoa1810858]
- OlympiAD Trial: Robson M, et al. “Olaparib for Metastatic Breast Cancer in Patients with a Germline BRCA Mutation.” NEJM (2017).
- FDA Label: Lynparza (olaparib) Prescribing Information. [FDA.gov Access]