Sourcing Disclosure: Licensed Bangladesh Global Sourcing Specialist facilitating global access under Named Patient Regulations.
Expert-reviewed clinical data for prescription-only medicine.
NSCLC Targeted Therapies: Biomarker Profiling and Precision Oncology Protocols
Introduction: The Shift to Precision Medicine
Non-Small Cell Lung Cancer (NSCLC) is the most common type of lung cancer, accounting for roughly 80% to 85% of all lung cancer diagnoses. For decades, the standard approach to treating NSCLC relied heavily on the physical appearance of the cancer cells under a microscope. If you had NSCLC, the treatment pathway was generally the same for everyone: surgery, radiation, or standard chemotherapy. It was a “one-size-fits-all” approach.
Today, medical science understands that NSCLC is not just one disease. At a cellular level, two patients with the exact same stage of NSCLC may have entirely different types of cancer driving their tumor growth. These differences lie in the cancer’s DNA. Precision medicine, also known as personalized medicine, involves analyzing the specific genetic makeup of your tumor to find the exact mutation causing the cancer to grow. Once identified, oncologists can often use “targeted therapies”—drugs designed specifically to attack that exact mutation, effectively shutting down the cancer’s ability to multiply. This method is the current standard of care recommended by organizations like the American Society of Clinical Oncology (ASCO) and the European Society for Medical Oncology (ESMO).
The Role of Biomarker Testing: Your First Step
Before starting any treatment for advanced NSCLC, biomarker testing (also called genomic testing, molecular testing, or tumor subtyping) is an essential first step. You cannot treat a specific mutation if you do not know it is there.
Biomarkers are specific genes, proteins, or other molecules found in or on cancer cells. By testing for these, your medical team can identify the genetic “driver” of your lung cancer. Testing is generally done in two ways: through a tissue biopsy or a liquid biopsy.
Tissue Biopsy
A tissue biopsy is the traditional method. A doctor extracts a small physical sample of the lung tumor using a needle, a bronchoscopy (a tube guided down the throat), or during surgery.
- Pros: It provides a large, reliable sample of the tumor, allowing pathologists to run comprehensive Next-Generation Sequencing (NGS) panels that look for dozens of mutations at once.
- Cons: It is an invasive procedure. Sometimes, tumors are located in hard-to-reach areas of the lung, making the biopsy difficult. Furthermore, tissue samples can take two to four weeks to analyze.
Liquid Biopsy
A liquid biopsy is a simple blood draw. When cancer cells die, they release tiny fragments of their DNA into the bloodstream, known as circulating tumor DNA (ctDNA). A liquid biopsy isolates and sequences this DNA.
- Pros: It is non-invasive, fast, and repeatable. Results often return in just seven to ten days. It is especially useful if a patient is too ill for a surgical biopsy or if the tumor is inaccessible.
- Cons: It is not always as sensitive as a tissue biopsy. If a tumor is not shedding enough DNA into the blood, the liquid biopsy might return a “false negative.”
Guidelines Note: Current ASCO guidelines recommend comprehensive biomarker testing for all patients with advanced NSCLC. Often, doctors will order both a tissue and a liquid biopsy simultaneously to gather information as quickly and accurately as possible.
Core Genetic Mutations in NSCLC
Scientists have identified several primary genetic mutations that drive NSCLC. For each of these, specific targeted therapies have been developed.
1. EGFR (Epidermal Growth Factor Receptor) Mutations
What is this mutation? EGFR is a protein found on the surface of cells that helps them grow and divide. In a healthy body, this protein receives signals telling the cell when it is time to multiply. An EGFR mutation means the gene is damaged, causing the receptor to be permanently stuck in the “on” position. This leads to rapid, uncontrolled cell division.
How common is it?
EGFR mutations are found in about 10% to 15% of NSCLC cases in the United States and Europe, but they are significantly more common in Asian populations, appearing in 40% to 50% of cases. They are most frequently found in patients who have never smoked or were light smokers.
Targeted therapy options:
Drugs called EGFR Tyrosine Kinase Inhibitors (TKIs) are the standard treatment. These drugs block the signal from the mutant EGFR protein, stopping the tumor from growing. Examples include:
- Osimertinib
- Erlotinib
- Gefitinib
- Afatinib
Drug Resistance:
Cancer is highly adaptable. Over time (usually 10 to 14 months with older generation drugs), the tumor may develop a new mutation to bypass the drug. The most common secondary mutation for early-generation EGFR inhibitors is the T790M mutation. When this occurs, the first drug stops working. Oncologists will then prescribe a third-generation TKI, such as Osimertinib, which is specifically designed to overcome the T790M resistance.
2. ALK (Anaplastic Lymphoma Kinase) Rearrangements
What is this mutation?
An ALK rearrangement occurs when the ALK gene breaks off and fuses with another gene (usually the EML4 gene). This fusion creates an abnormal protein that constantly signals the cancer cells to grow and spread.
How common is it?
ALK fusions are found in about 3% to 5% of NSCLC patients. It typically affects younger patients (often under the age of 50) and those with a history of light or no smoking.
Targeted therapy options:
ALK inhibitors are highly effective and can often shrink tumors significantly, including tumors that have spread to the brain. Standard options include:
- Alectinib
- Brigatinib
- Lorlatinib
- Crizotinib
Drug Resistance:
Like EGFR, ALK-positive cancers eventually find workarounds, mutating their structure so the drug cannot bind to the protein. When an older drug like Crizotinib stops working, doctors move to next-generation inhibitors like Alectinib or Lorlatinib, which are stronger and better at crossing the blood-brain barrier.
3. KRAS (Kirsten rat sarcoma viral oncogene homolog) Mutations
What is this mutation?
The KRAS gene normally acts as an internal switch that controls cell growth. When mutated, the switch is jammed “on.” For decades, KRAS was considered “undruggable” because the protein’s physical shape is very smooth, making it difficult for drugs to attach to it.
How common is it?
KRAS is the most common mutation in NSCLC, found in about 25% to 30% of patients. It is strongly associated with a history of smoking. The most common specific subtype of this mutation is KRAS G12C, which makes up about 13% of all NSCLC cases.
Targeted therapy options:
Recent medical advancements have finally produced drugs that can target the KRAS G12C mutation specifically by fitting into a tiny, newly discovered groove on the protein.
Drug Resistance:
Because these therapies are relatively new, researchers are still mapping exactly how resistance develops. Often, the cancer activates alternative growth pathways (like the MEK pathway). Clinical trials are currently testing combinations of KRAS inhibitors with other drugs to delay this resistance.
4. ROS1 Rearrangements
What is this mutation?
Similar to ALK, the ROS1 gene can fuse with a nearby gene, creating a rogue protein that drives rapid cancer growth. It behaves very similarly to the ALK mutation.
How common is it?
This is a rare mutation, occurring in only 1% to 2% of NSCLC cases. It shares patient demographics with ALK, presenting mostly in younger, non-smoking patients.
Targeted therapy options:
Because ROS1 and ALK proteins are structurally similar, some drugs work for both.
- Entrectinib
- Crizotinib
- Repotrectinib
Drug Resistance:
When resistance occurs (often through a secondary mutation like G2032R), patients may be transitioned to newer, stronger inhibitors like Lorlatinib or enrolled in clinical trials for emerging therapies.
5. BRAF V600E Mutations
What is this mutation?
The BRAF gene produces a protein that is part of a signaling pathway (the MAPK/ERK pathway) that regulates cell division. A specific error called the V600E mutation causes this protein to become overactive. This mutation is also famously associated with melanoma (skin cancer).
How common is it?
BRAF mutations appear in 1% to 3% of NSCLC patients.
Targeted therapy options:
Treatment usually involves combining two drugs. One blocks the BRAF protein, and the other blocks MEK, a related protein in the same pathway. By blocking two points on the same pathway, the treatment is much more effective.
- Encorafenib (BRAF inhibitor) + Binimetinib (MEK inhibitor)
Drug Resistance:
Resistance mechanisms for BRAF are complex, often involving the cancer finding a completely different signaling pathway to use. When targeted therapy stops working, the next step is usually traditional chemotherapy, immunotherapy, or clinical trials.
6. MET Exon 14 Skipping Mutations
What is this mutation?
The MET gene provides instructions for making a protein that helps cells signal to each other. In this mutation, a specific piece of the genetic code (Exon 14) is accidentally skipped or deleted during the cell’s copying process. This results in a defective protein that triggers continuous cell growth.
How common is it?
It is found in 3% to 4% of NSCLC cases and is more common in older adults, averaging around age 70.
Targeted therapy options:
Highly specific MET inhibitors have recently been approved to treat this mutation.
- Capmatinib
- Tepotinib
Drug Resistance:
Resistance can develop through further mutations within the MET gene itself or through the activation of other genes like KRAS. Next steps involve alternative targeted drugs or systemic chemotherapy.
Special Focus: How Targeted Therapies Work Differently Than Chemotherapy
To understand the value of precision medicine, it is helpful to compare it to traditional treatments.
Standard chemotherapy is systemic and cytotoxic, meaning it travels throughout the entire body and kills any cells that are dividing quickly. Because cancer cells divide rapidly, they are heavily impacted. However, many healthy cells in your body also divide quickly—such as the cells in your hair follicles, the lining of your stomach, and your bone marrow. Chemotherapy attacks these healthy cells too, which is what causes common side effects like hair loss, severe nausea, and a weakened immune system.
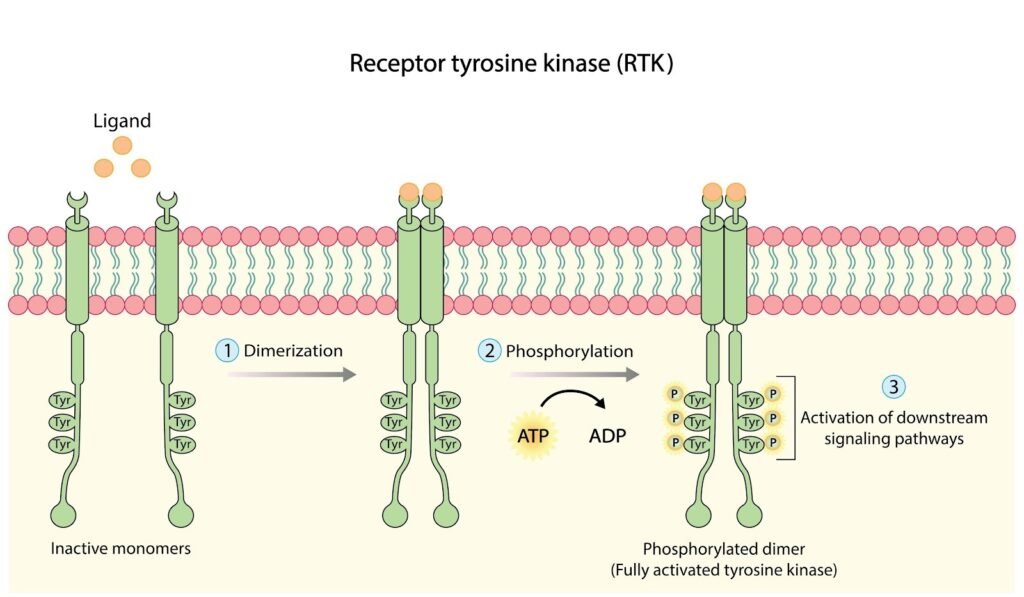
Targeted therapies, primarily small-molecule drugs called Tyrosine Kinase Inhibitors (TKIs), work differently. They are cytostatic, meaning they block tumor cell proliferation rather than directly killing the cell.
TKIs are designed to recognize and attach only to the specific mutated proteins (kinases) on the cancer cells. Think of the mutated protein as a lock, and the targeted therapy as a highly specific key. When the TKI binds to the mutated protein, it blocks the chemical signals the cancer cell needs to divide and survive. Because these drugs focus primarily on the mutated cells and largely ignore normal, healthy cells, they generally cause less collateral damage to the rest of the body compared to chemotherapy.
(Note: While some multi-targeted TKIs like Pazopanib are used in other cancers like soft tissue sarcoma, NSCLC relies on highly specific TKIs engineered for the precise lung cancer mutations mentioned above).
Managing Side Effects at Home
While targeted therapies avoid many traditional chemotherapy side effects, they are not side-effect-free. Because some normal cells (like those in the skin and gut) share similar proteins to the ones being targeted, patients often experience specific issues. Proactive management is essential for maintaining a good quality of life.
1. Skin Rashes (Acneiform Rash)
This is the most common side effect of EGFR inhibitors. It looks like acne but is not treated like acne. It usually appears on the face, chest, and back.
- Management: Do not use over-the-counter acne medications (like salicylic acid or benzoyl peroxide), as these will dry out and severely irritate the skin.
- Hygiene: Wash your skin with lukewarm water and mild, soap-free cleansers.
- Moisturize: Apply thick, alcohol-free, fragrance-free moisturizing creams (not lotions) twice a day.
- Protection: These drugs make your skin highly sensitive to the sun. Wear broad-spectrum SPF 30+ sunscreen every day, even indoors or on cloudy days, and wear a wide-brimmed hat outdoors. If a rash becomes painful or infected, your oncologist can prescribe topical steroids or oral antibiotics.
2. Diarrhea and Digestive Issues
Many TKIs, particularly ALK and EGFR inhibitors, irritate the lining of the digestive tract.
- Dietary Adjustments: Eat small, frequent meals rather than large ones. Adopt a low-fiber, bland diet when symptoms flare up (the BRAT diet: Bananas, Rice, Applesauce, Toast). Avoid spicy, greasy, or heavily dairy-based foods.
- Hydration: Severe diarrhea can lead to dehydration and loss of electrolytes. Drink plenty of clear fluids, broths, and oral rehydration solutions.
- Medication: Keep over-the-counter anti-diarrheal medications (like Loperamide) at home, but ask your doctor for specific dosing instructions before starting treatment.
3. Fatigue
Targeted therapies require your body to process strong medications continually, leading to profound tiredness that does not go away with sleep.
- Activity: It seems counterintuitive, but light, consistent physical activity (like a 20-minute daily walk) is proven to reduce cancer-related fatigue better than prolonged bed rest.
- Pacing: Plan your day to include rest periods. Do not overexert yourself on days you feel energetic, as it can cause a “crash” the next day.
4. Liver Enzyme Elevations
Many targeted drugs are processed by the liver. Sometimes, the drugs can cause inflammation, which shows up as elevated liver enzymes on a blood test.
- Monitoring: You will not usually feel this side effect, which is why routine blood tests every few weeks are mandatory.
- Action: Avoid alcohol and do not take any new supplements or herbal remedies without telling your doctor, as these can further stress the liver and interact dangerously with your cancer medication.
Patient-Doctor Communication: Questions You Must Ask
Being an active participant in your treatment plan improves outcomes. Print this list and take it to your next oncology appointment:
- Has my tumor been comprehensively tested for all actionable biomarkers using Next-Generation Sequencing (NGS)?
- Were both a tissue biopsy and a liquid biopsy performed? If not, why?
- What specific genetic mutation is driving my cancer?
- Based on my mutation, what is the most effective targeted therapy currently approved by the FDA/EMA?
- What are the most common side effects of this specific drug, and what prescriptions can I take home today to manage them?
- Are there any foods, supplements, or other medications I need to avoid while taking this targeted therapy?
- How frequently will I need scans and blood tests to monitor if the drug is working?
- How will we know if the cancer is developing resistance to this drug?
- If this treatment eventually stops working, what is our “Plan B” or next-line therapy?
- Are there any clinical trials evaluating new treatments for my specific mutation that I qualify for right now?
Future Outlook & Clinical Trials
A common misconception is that clinical trials are an experimental “last resort” for patients who have run out of options. In modern oncology, this is false.
Clinical trials are a valid, front-line treatment option. Because precision medicine is advancing so rapidly, the standard of care changes every year. By joining a clinical trial, patients often gain access to tomorrow’s breakthrough targeted therapies years before they are available to the general public.
Researchers are currently testing fourth-generation TKIs, combining targeted drugs with immunotherapy, and finding ways to target mutations (like certain KRAS variants) that currently have no approved drugs. If your doctor suggests a clinical trial, it means they believe the trial offers a drug that may be superior to the current standard treatment.
Conclusion
A diagnosis of Non-Small Cell Lung Cancer is daunting, but the landscape of treatment has fundamentally changed. By insisting on comprehensive biomarker testing, you ensure that your treatment is tailored specifically to the genetic code of your tumor. Targeted therapies offer the ability to control the disease for extended periods with a focus on maintaining a high quality of life. Work closely with your medical team, manage side effects proactively, and stay informed about your specific diagnosis.
Frequently Asked Questions (FAQ)
Can my genetic mutation change over time?
Yes. As the cancer is treated, the tumor cells adapt and can develop new, secondary mutations to survive the drugs. This is why repeated biopsies (often liquid biopsies) are done when a drug stops working.
Are these lung cancer mutations inherited from my parents?
No. The mutations that drive NSCLC (like EGFR, ALK, KRAS) are called “somatic” mutations. This means they are acquired during your lifetime due to environmental factors or random cell copying errors. They are not passed down to your children.
How long do targeted therapies work?
It varies widely depending on the mutation, the specific drug, and your individual biology. Some patients develop resistance within a year, while others remain stable on the same targeted therapy for many years.
Do I still need chemotherapy if I take targeted therapy?
Usually, no. For patients with a targetable mutation, TKIs are used as the first line of defense instead of chemotherapy. Chemotherapy is generally held in reserve for if or when the targeted therapies and their subsequent generations stop working.
Why does my doctor want me to stop taking grapefruit juice?
Grapefruit and Seville oranges contain compounds that interfere with an enzyme in your digestive tract that helps process many targeted therapies. Consuming them can cause the drug levels in your blood to spike to toxic levels. Always ask your pharmacist about food interactions with your specific TKI.
Source of Researche:
https://pubmed.ncbi.nlm.nih.gov
Notice to Healthcare Professionals: Oncology is a rapidly evolving field. Our editorial team welcomes peer feedback, clinical insights, and research updates from credentialed medical professionals. Contact our medical board to share your expertise.
The content, clinical monographs, and informational resources provided on genericoncology.com are for educational and informational purposes only. While our content is rigorously evaluated and vetted by international medical reviewers to ensure clinical accuracy, this website does not provide medical diagnoses, treatment plans, or professional medical advice. Accessing this website, reading its content, or contacting our support team does not establish a doctor-patient relationship. You must always consult with your primary oncologist or a qualified healthcare provider before initiating, altering, or discontinuing any targeted therapy or medical treatment.
Medically Reviewed by Dr. Salma Elreedy ,Clinical Ocncology, Sphinx Cure Oncology Center