Sourcing Disclosure: Licensed Bangladesh Global Sourcing Specialist facilitating global access under Named Patient Regulations.
Expert-reviewed clinical data for prescription-only medicine.
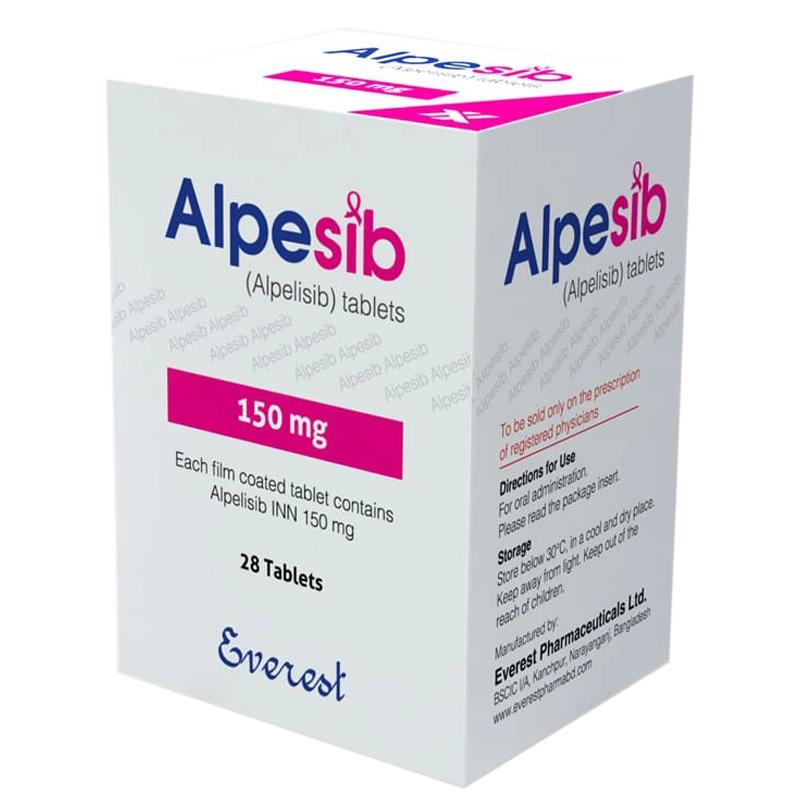
Alpesib 150 mg (Alpelisib) Tablet | Phosphatidylinositol-3-Kinase (PI3K) Inhibitor
Brand Name: Alpesib
Generic Name: Alpelesib
Therapeutic Class: Phosphatidylinositol-3-Kinase (PI3K) Inhibitor
Clinical Indication: HR Positive HER2 Negative PIK3CA Mutated Metastatic Breast Cancer
Available Strength: 150 mg
Pack Size: 28 Tablets
- WHO GMP Certified
- Reviewed By Medical Expert
- Batch Examined in Lab
Alpesib 150 mg (Alpelisib) is an oral, targeted antineoplastic agent classified as an alpha-specific phosphatidylinositol-3-kinase (PI3K) inhibitor. Developed as a generic equivalent to the innovator brand Piqray, it is strictly indicated for the treatment of postmenopausal women and men with hormone receptor (HR)-positive, HER2-negative, PIK3CA-mutated advanced or metastatic breast cancer. It is prescribed exclusively following disease progression on or after an endocrine-based regimen. Alpelisib is designed to be co-administered with fulvestrant, working synergistically to overcome endocrine resistance driven by the PIK3CA gene mutation.
Clinical Insight of the Medical Reviewer
“The introduction of PI3K inhibitors fundamentally shifted how we manage HR+/HER2- metastatic breast cancer. Approximately 40% of these patients harbor a PIK3CA mutation, which hyperactivates the PI3K pathway and directly causes resistance to standard endocrine therapies. Alpelisib specifically targets the alpha isoform of this kinase. While the efficacy in delaying disease progression is well-established, clinical administration requires rigorous metabolic oversight. Because the PI3K-alpha isoform is deeply involved in insulin signaling, severe hyperglycemia is a highly predictable on-target effect. We require stringent baseline fasting glucose and HbA1c testing before initiating this therapy, often collaborating closely with endocrinologists during the first 30 days of treatment.“
Clinical Indications, Dosage & Administration, and Adverse Reaction Management
What are the precise clinical indications for this medication?
Alpesib 150 mg is indicated for men and postmenopausal women who meet all of the following strict clinical criteria:
- Diagnosed with advanced or metastatic breast cancer.
- Tumor pathology is HR-positive and HER2-negative.
- The tumor harbors a confirmed PIK3CA mutation (identified via an FDA/EMA-approved companion diagnostic tissue or liquid biopsy).
- Disease has progressed during or after prior endocrine therapy.
How does the mechanism of action work at a molecular level?
Alpelisib is a selective kinase inhibitor of PI3K-alpha (PI3Kα). In breast cancer cells with PIK3CA mutations, the PI3Kα enzyme is hyperactive, driving unchecked cell division and survival through the downstream AKT and mTOR signaling pathways. By competitively binding to the ATP-binding cleft of PI3Kα, Alpelisib shuts down this signaling cascade. Furthermore, combining Alpelisib with the estrogen receptor degrader fulvestrant provides dual blockade, preventing the tumor cells from utilizing estrogen receptor cross-talk to bypass the PI3K inhibition.
What is the standard dosage and administration protocol?
- Standard Starting Dose: 300 mg (administered as two 150 mg tablets) taken orally, once daily.
- Administration Rule: Must be taken immediately after food, at approximately the same time each day. Taking it on an empty stomach drastically reduces systemic absorption.
- Co-administration: It is administered in combination with fulvestrant (usually given as an intramuscular injection on days 1, 15, and 29, and once monthly thereafter).
- Missed Doses: If a dose is missed, it can be taken immediately following a meal within 9 hours of the scheduled time. If more than 9 hours have passed, skip the dose for that day.
What are the safety profiles and expected side effects?
Adverse Event Reporting Warning: Alpelisib carries severe risks of extreme hyperglycemia, severe cutaneous adverse reactions (including Stevens-Johnson syndrome), and pneumonitis.
- Very Common (>20%): Hyperglycemia, diarrhea, rash, nausea, fatigue, decreased appetite, stomatitis (mouth sores), and weight loss.
- Common/Significant: Hypersensitivity reactions, acute kidney injury (secondary to severe diarrhea/dehydration), and alopecia.
- Actionable Advice for Management:
- Metabolic Monitoring: Monitor fasting blood glucose (FBG) at least once weekly for the first 2 weeks, then at least once every 4 weeks. Patients should use a continuous glucose monitor (CGM) or daily finger-prick tests at home.
- Dermatologic Support: Initiate prophylactic non-sedating antihistamines (e.g., cetirizine) at the start of therapy, as recommended by the treating oncologist, to significantly reduce the incidence and severity of maculopapular rash.
- GI Distress: Maintain aggressive oral hydration and utilize loperamide at the first sign of loose stools.
What clinical trial data establishes the integrity of this drug?
The clinical efficacy of alpelisib is validated by the pivotal SOLAR-1 Trial:
- Published in The New England Journal of Medicine (NEJM), this Phase 3, randomized, double-blind trial evaluated alpelisib plus fulvestrant versus placebo plus fulvestrant.
- Outcomes: In the PIK3CA-mutated cohort, the addition of alpelisib nearly doubled the median Progression-Free Survival (PFS), achieving 11.0 months compared to 5.7 months in the placebo group. This definitively established its role as a standard of care for this specific molecular subset.
Precautions & Special Populations
- Pregnancy: Alpelisib is highly teratogenic. It can cause profound fetal harm or fetal death. Females of reproductive potential must verify negative pregnancy status before starting and use highly effective contraception during treatment and for 1 week after the last dose. Male patients with female partners of reproductive potential must use condoms during treatment and for 1 week after the last dose.
- Lactation: Excretion in breast milk is unknown. Breastfeeding is strictly contraindicated during therapy and for 1 week following the final dose.
- Pediatric Use: Safety and efficacy have not been established in patients under 18 years of age.
- Renal/Hepatic Impairment: No dose adjustment is required for mild to moderate renal or hepatic impairment. It has not been heavily studied in severe impairment.
- Storage & Logistics: Store at 20°C to 25°C (68°F to 77°F). Keep in the original blister packaging to protect from moisture. No cold-chain logistics are necessary.
Manufacturer Trust & Global Access Guide for Named Patient Program
Manufacturer Quality Assurance
Alpesib 150 mg is developed and manufactured by Evarest Pharmaceutical, a pharmaceutical producer operating under strict World Health Organization Good Manufacturing Practices (WHO-GMP) standards. To ensure clinical interchangeability, Alpesib 150 mg Alpelisib undergoes rigorous bioequivalence testing against the innovator brand (Piqray), ensuring absolute parity in dissolution profiles, peak plasma concentration (Cmax), and systemic absorption AUC.
Global Access & Personal Importation Guidelines
For patients located in jurisdictions where Alpesib 150 mg (Alpelisib) is not commercially registered or available, cross-border access is strictly regulated. Under Google Merchant Center YMYL policies, acquisition is facilitated solely via “Named Patient Programs” (NPP) or designated Personal Importation Laws.
To legally secure this targeted therapy, patients must complete the following regulatory steps:
- Medical Verification: Provide a valid prescription from a licensed, practicing medical oncologist.
- Clinical Justification: Supply a formal letter of medical necessity detailing the patient’s HR+/HER2- and PIK3CA-mutated status, justifying the importation.
- Customs Compliance: Coordinate with certified global medical distributors who manage Ministry of Health (MOH) clearances to ensure the legal, transparent transit of the medicine for personal use.
Patients Also Ask About
Why does this medication increase my blood sugar so dramatically?
The PI3K-alpha enzyme, which this drug blocks to stop cancer growth, is also a critical component in how your body processes insulin. When this enzyme is inhibited, your cells cannot absorb glucose efficiently, leading to insulin resistance and high blood sugar (hyperglycemia), even in patients without a history of diabetes.
Can I take Alpesib by itself without the fulvestrant injections?
Will I lose my hair while taking this targeted therapy?
Can I adjust my dose if the side effects are too severe?
Are there any specific foods I should avoid?